Daytrana Patch Skin Problems: Lawsuits Reviewed for Discoloration, Scaring
Following recent FDA warnings about the risk of skin problems from the Daytrana ADHD patch, the product liability lawyers at Saiontz & Kirk, P.A. are reviewing whether users who have experienced skin discoloration, scaring or disfigurement may be entitled to financial compensation through a Daytrana patch lawsuit.
Daytrana (methylphenidate transdermal system) is a medicated patch applied once per day for the treatment of attention deficit and hyperactivity disorder (ADHD). It was originally approved by the FDA in 2006, and has been plagued with problems and Daytrana recalls.
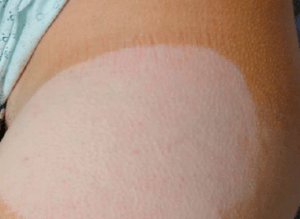
Following reports of users experiencing loss of skin pigmentation, the FDA issued a drug safety communication last month warning about a risk of chemical leukoderma from Daytrana side effects.
This potential side effect of Daytrana may cause individuals to experience:
- Changes to Skin Color
- Complete Loss of Skin Pigmentation
- Light or White Patches of Skin
While chemical leukoderma does not cause any physical harm, it usually results in permanent scarring or disfigurement, which can cause severe embarrassment, emotional distress or otherwise impact an individuals self-image or quality of life.
Daytrana Chemical Leukoderma Warnings
The FDA indicates that all reported cases of chemical leukoderma from Daytrana have been permanent, resulting in lost pigmentation that stretched up to eight inches across for some users.
While most of the Daytrana skin patch problems appeared where the ADHD patch was regularly applied or rotated, in some cases the lost skin color developed elsewhere on the body. The problem can appear as soon as a few months after first Daytrana patch use, or can wait several years to appear.
Prior to the recent label change announced by the FDA, it appears that the Daytrana patch warnings failed to adequately warn users and the medical community about the risk of these permanent and disfiguring side effects, which may impact children or adults using the ADHD skin patch.
It appears likely that most users would have elected not to wear the Daytrana patch for ADHD if they had been aware of the risk of permanently disfiguring skin problems, as many other treatment options are available for the condition.
Daytrana Patch Class Action Lawyers
The Daytrana lawyers at Saiontz & Kirk, P.A. are reviewing individual chemical leukoderma lawsuits, as well as potential Daytrana class action lawsuits as a result of the drug maker’s failure to adequately warn about the risk of skin problems.
It appears that the manufacturers may have placed their desire for profits before consumer safety, exposing individuals to a risk of severe skin problems from Daytrana that could have been avoided if adequate warnings had been provided.
To submit information about scarring or skin color changes that may have been caused by Daytrana patch side effects, request a free consultation and claim evaluation.


6 Comments • Add Your Comments
Kevin says:
I have used the Daytrana patch since 2006 and have had no problems. It is the best ADHD med I have found, especially because of its delivery system. You lawyers need to back off…I doubt seriously there are many bona fide cases of what you describe, other than a few looking to make a fast buck.
Posted on August 2, 2015 at 5:15 pm
Julie says:
I have been fighting for my daughter since we started to notice what appeared to be a spot the size of a quarter appear on her hip in the same spot where she placed the patch. I contacted Daytrana at the time and they were no help. The spot became larger over time to the size of a 50 cent piece and I decided to take her off. She was on the patch 2 years. Strangely, the area increased in size more rapidly once the patch was gone it increased past her hip to the the back of her buttocks an is larger than a dollar bill. It is very noticeable when wearing a bathing suit and causes my daughter, now a teenager distress whenever she has to be around her peers in a bathing suit because it doesn’t tan and is very noticeable. We went to the dermatologist which diagnosed it as chemical induced hypo pigmentation. It is permanent, will never go away, is unpredictable meaning it could start growing again or other spots may appear years later. the area is highly susceptible to skin cancer. My daughter used to have Olive skin like I do. When she was 5 and 8 years old and we would go to Hawaii she would always get a really nice tan and would not burn hardly at all. This year on our trip to Hawaii we notice it was difficult for her to tan and she would burn right away. My daughters skin has been altered. Her life has now been permantley changed. At one point I contacted the FDA and gave them all my documentation and pictures of my daughter. 6 months ago they contacted me to inform me because of the information I sent in and the information from other, the reversed the warning label. They also asked for my permission to use my daughters photo in a medical journal for a report the wrote referencing my information and my daughters picture. I’m not a person that is sue happy. Matter of fact I have never sued anyone. However, my daughter’s life has been changed now and not just cosmetically, physically. She will forever have to be limited to her sun exposure in the summer. she will need to go to the dermatologist 2 times a year to check her pigment. She has the expense of spray tanning that area to blend it so not to get ridiculed by other teens that are insensitive. And on top of it she has anxiety over it now. There is a new symptom we are having checked out that has to do with her skin as well and that is with the contact of water and her hands. Novum I’m sure knew about these possible side affects. My daughter was one of the first to try it because like many, couldn’t swallow a pill. Had I known there was a risk with losing pigmentation permanently I would have thought twice and researched alternatives. Something permanent is much different than possible damage. Yes, the patch works great and has many benefits however, the risk of to losing something permanent are not with it. The other severe thing was my daughter looked like a skeleton because it cause so much appetite loss. That is also a bad side affect. I really think Novum needs to have some sort of action brought against them for this berfore too many people go thru what my daughter is. It took 2 years for this thing to really appear to a 50 cent size and 3 years to a dollar bill size.
Posted on April 17, 2016 at 1:04 pm
amy says:
My son has been on Daytrana for 2 1/2 years. NO discoloration from the patches. They cant predict that some will have this happen. This drug is the only drug that works for my son because he can not swallow pills. It is the ONLY drug that last. It is the ONLY drug that does not kill his appetite. If anything they should be sued for taking them away and holding off distribution. Do you know what these kids go through finding a drug that actually works? Then they do a voluntary recall and hold off for months to start redistributing them. My son had to go back on Focalin at the same time he had to have a bone graft surgery. Focalin only last a few hours. Insurance will not cover 2 pills a day. He was finally gaining weight and not looking like a bag of bones and now I have to beg him to eat. I just cant wait to see his grades when school starts. PLEASE put Daytrana back on the market before I lose what sanity I have left.
Posted on August 2, 2016 at 6:34 pm
charles says:
Julie: My son had been on the patch for 4 years before we abruptly took him off for loss of pigmentation. Unfortunately for us he is spotted on both hips, under his neck, and in private areas. Spots began small but have continued to grow in size. We are seeing a specialist in Boston and will be filing suit. Please contact me if you want to be part of the suit. This is a nightmare for my 16 almost 17 year old and will be a permanent life-time disfigurement.
Posted on January 3, 2017 at 6:13 pm
Kerrie says:
I too have been having the same issues for years now with my daughter who is now 16. She started with the Daytrana patch in 2011 and only used it for a few months before noticing the skin color change. She now has a spot the size of an egg on her hip that has no skin color and is very noticeable while wearing a bathing suit. As a 16 year old, this is so distressing for her. I have had a difficult time finding an attorney or a class action suit that will take this on. Any help you can offer would be appreciated
Posted on June 9, 2017 at 10:43 am
Kerrie says:
My daughter has suffered with this same problem for years now. In the summer its very uncomfortable for her because it is so noticeable on her hip (where the doctor said it should have been placed!) We have had no luck finding any legal help. Seems no one wants to take on the pharmaceutical companies. What are we supposed to do, just accept it? It is SO wrong in every way and someone needs to be held accountable. Have you had an success in finding an attorney?
Posted on September 14, 2017 at 1:47 pm