Cartiva Toe Implant Lawsuit
Lawsuits are being pursued against the manufacturers of the Cartiva synthetic cartilage implant for individuals throughout the United States who suffered complications that resulted in the need for a toe fusion, revision or replacement surgery. Cartiva lawsuit payouts may be available as a result of design defects that resulted in a high toe implant failure rates.
The Cartiva synthetic cartilage device received initial FDA approval in 2016, and was marketed by the manufacturer as a breakthrough alternative to the existing fusion surgery technique to treat big toe arthritis.
The Cartiva toe implant surgery was intended to increase mobility of the foot, and improve overall quality of life. However, patients began experiencing much higher rates of pain and complications from Cartiva implant failures than were previously disclosed by the manufacturer during clinical trials.
While Cartiva was approved largely on the perceived benefits and clinical trials that showed only a 13.5% toe implant failure rate, a study was published in 2020 finding the actual rate of Cartiva implants failing in patients within the first two years were as high as 79%.
Despite mounting evidence highlighting increased rates of failure, it was not until October 31, 2024 that Stryker issued a Cartiva implant recall based on recently published data and post market reports indicating that patients who received a Cartiva implant experienced higher rates of revision, removal, implant subsidence, displacement, pain, nerve damage or fragmentation than previously disclosed.
As a result of the manufacturers failure to adequately research the safety and effectiveness of Cartiva, our product liability lawyers are now pursuing Cartiva implant failure lawsuits for individuals across the country who have suffered injuries or required revision or replacement surgery.

Saiontz & Kirk, P.A. is investigating Cartiva toe implant lawsuits for individuals who meet the following criteria;
- Received a Cartiva big toe implant; AND
- Experienced significant complications that resulted in the need for subsequent toe surgery, which has either been performed, scheduled or recommended.
The product liability lawyers at Saiontz & Kirk, P.A. provide free consultations and case evaluations to help determine if Cartiva lawsuit settlement benefits may be available for you or a loved one.
2025 Cartiva Lawsuit Update
Following studies linking the Cartiva implant to high failure rates, a growing number of individuals are now pursuing Cartiva big toe joint implant lawsuits, alleging that defective design resulted in severe pain and the need for revision or replacement surgery.
- The Cartiva big toe joint implant was approved as a Tier III medical device in 2016, for treatment of big toe arthritis.
- The manufacturer aggressively advertised the Cartiva implant after it was introduced as a safe alternative to big toe fusion surgery.
- Physicians and patients have reported significantly higher rates of the Cartiva implant failures than previously reported in clinical trials by the medical device company.
- Stryker issued a Cartiva implant recall on October 31, 2024, publicly recognizing the increased risk of failure rates and need for revision, replacement surgery.
- Individuals are now pursuing Cartiva lawsuits against the manufacturer due to the pain caused by the device failing, and the need for additional surgery.
- Lawyers are actively pursuing Cartiva implant failure lawsuits for individuals who have experienced implant complications resulting in the need for joint removal surgery.
Is There a Cartiva Toe implant Recall?
Yes. Stryker issued a Cartiva implant recall on October 31, 2024, after a review of recent data and postmarket reports highlighted that the rates of complications in patients with the Cartiva SCI are significantly higher than initially reported during its 2016 Premarket Approval (PMA).
According to the Cartiva implant recall notice, these complications require implant revision or removal and include subsidence, displacement, persistent pain, nerve damage, and fragmentation. The frequency of these incidents requiring surgical intervention has surpassed the rates initially reported in the premarket and post-approval studies.
The recall included all batches of the Cartiva Synthetic Cartilage Implant produced in 6 millimeter, 8 millimeter, 10 millimeter, and 12 millimeter sizes, distributed from July 2016 through October 2024. The affected products bear the following catalog and primary device identifier (PDI) numbers:
- CAR-06-US (6mm) – PDI 00852897002328
- CAR-08-US (8mm) – PDI 00852897002021
- CAR-10-US (10mm) – PDI 00852897002038
- CAR-12-US (12mm) – PDI 00852897002335
As part of the recall, healthcare providers are directed to monitor patients for symptoms of Cartiva implant failure, which can include pain, difficulty walking, skin reactions, stiffness, swelling, or weakness in the big toe joint.
Individuals experiencing adverse reactions or quality issues with the implant should immediately report these incidents to Stryker’s Trauma & Extremities division complaint department via email, as well as to the FDA’s MedWatch Serious Injury Reporting Program.
What is the Cartiva toe implant and what are its uses?
The Cartiva toe implant is a small, gel-like device that is made of a biomaterial that mimics natural cartilage in the big toe joint.
The Cartiva synthetic cartilage joint is used to replace damaged or deteriorated cartilage in the first metatarsophalangeal joint, to provide cushioning and prevent the joint bones from rubbing together.
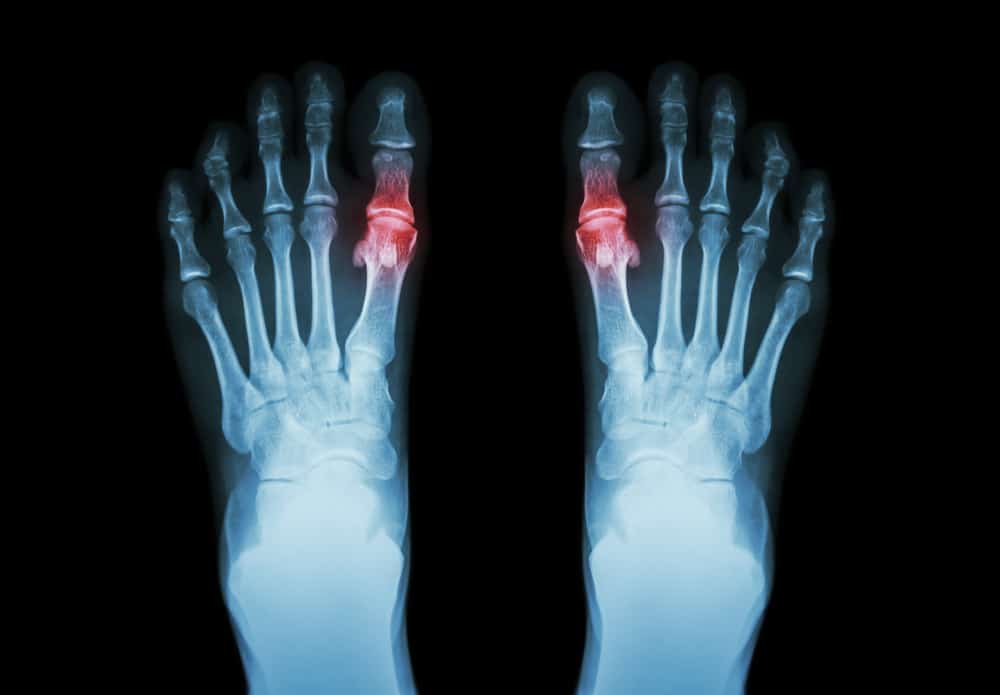
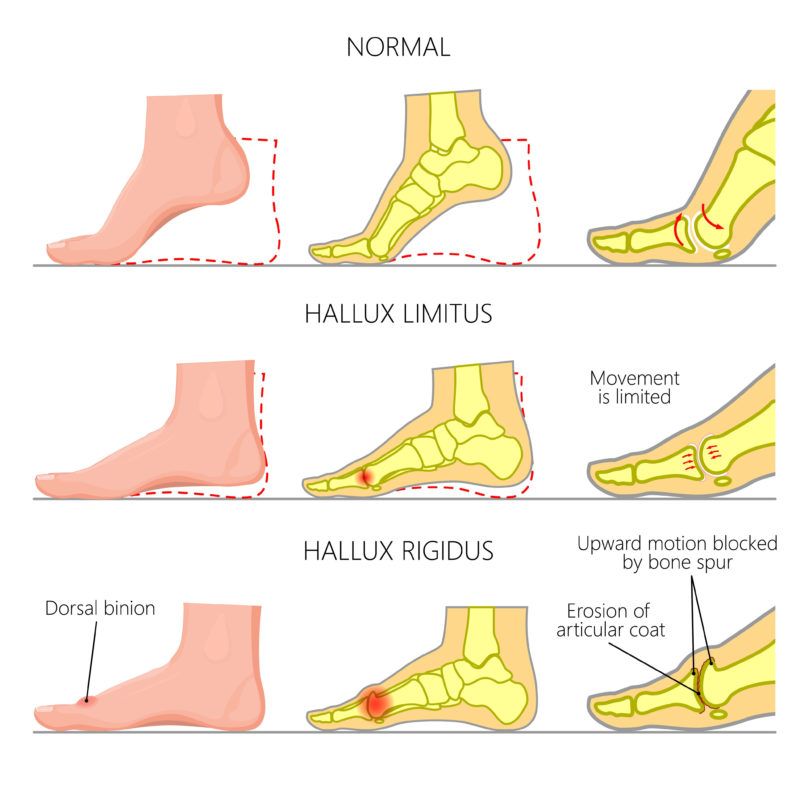
Cartiva implants are routinely offered to individuals who suffer from conditions known as Hallux Limitus or Hallux Rigidus, which are forms of big toe arthritis that cause debilitating pain and stiffness in the joint that can worsen over time and limit the mobility of the big toe.
Hallux Limitus is a form of degenerative arthritis that causes a limited range in motion of the big toe.
Symptoms of Hallux Limitus can include difficulty walking normally, pain on weight bearing surfaces, discomforts while turning or sudden movements and often there may be bony growths in the area. However, hallux limitus may progress to hallux rigidus.
Hallux Rigidus is a severe type of degenerative arthritis in which hardly any or no cartilage of the joint remains, and causes a much more painful restricted range in motion of the toe.
Symptoms of Hallux Rigidus can range from difficulty bending the great toe joint when walking to intense pain and stiffness in the joint itself. Stiffness tends to worsen over time, with advanced cases becoming unable to move their toe at all.
Cartiva Implant Failure
Cartiva was first given premarket approval by the FDA in 2016, based on the results of the Motion clinical study, which the manufacturer used to establish a “substantial equivalence” to arthrodesis, and claimed there was only a 13.5% Cartiva implant failure rate.
However, the results of this Cartiva study have never been duplicated and the failure rate is much higher than the Motion study predicted, according to follow-up studies and case reports. In fact, there is evidence that revisions after a Cartiva failure may cause more severe long term foot side effects than originally receiving arthrodesis.

Cartiva Device Associated With 64% Failure Rate after 4 Weeks: Study
According to a study published by the American Orthopedic Foot and Ankle Society in November 2020, researchers found as many as 64% of patients who received Cartiva for Hallux Rigidus experienced device failure within four weeks of the surgery. The number of Cartiva implant failures increased to 79% at 19 months post-surgery.
Specifically, the study found the most commonly reported reason for Cartiva failure was subsidence, which is the sinking of the implant into the bone. This condition has been associated with severe toe pain that often requires revision surgeries.
Additional side effects of Cartiva found in the study included:
- 40% of patients experienced erosion of the toe bone that is closest to the metatarsals,
- 57% showed radiologic lucency around the implant,
- 43% of patients reported no pain or mobility improvement 19 months after surgery.
Half of Cartiva Recipients Experience Stiffness and Limited Mobility
In a study published in the medical journal Cureus on November 19, 2023, researchers from the U.K. found that 45.5% of Cartiva recipients experienced complications related to the medical procedure, surgical process, and the implant itself. Additionally, a 55% reported persistent pain at their final follow-up appointment while 50% experienced stiffness or limited mobility, and 10% reported persistent swelling.
The study also pointed out that ongoing pain was a frequent issue, leading to the need for additional interventions such as orthotics, debridement, or repositioning of the implant. Researchers found that nearly 20% of individuals required subsequent surgeries to correct or remove the implant.
Nearly 30% of Cartiva Implant Patients Needed Further Surgery
The October 2024 issue of Foot and Ankle Surgery presented a study by T.L. Lewis et al., comparing the Cartiva Synthetic Cartilage Implant (SCI) with arthrodesis (joint fusion) for hallux rigidus treatment.
This retrospective cohort study involved 33 patients and revealed that 29.4% of those with the Cartiva implant required reoperation, significantly higher than the 0% reoperation rate seen in the joint fusion group.
Cartiva Implant Failures Worsen Patient Outcomes
While Cartiva implants have been marketed as a safe and beneficial alternative to big toe fusion surgery, medical experts have challenged these claims, suggesting that when a Cartiva implant fails, it may actually cause a patient more harm than initially receiving a toe fusion.
The Mayo Clinic reported salvage surgery to repair a failed Cartiva implant creates significant reconstructive problems because joint replacement surgery requires a large amount of bone to be removed.
As a result, reports suggest many are left with a significantly shortened great toe that can lead to a condition known as transfer metatarsalgia. This condition creates excessive offloading and typically causes the big toe to stick up, not touching the floor.
Why Does The Cartiva Implant Fail?
Medical experts from the University Foot and Ankle Institute have offered several theories on why the Cartiva synthetic cartilage implant can fail, all of which revolve around a suggested faulty and defective design that makes them prone to loosening and allowing the gel-like device to slip into the bone, known as subsidence.
Improperly Textured Design
Medical experts theorized that Cartiva implants were improperly designed with a smooth surface on the surrounding sides, rather than containing some form of irregularity, which may allow them to move or sink out of place.
The University Foot & Ankle Institute has suggested this exceptionally smooth design creates a functional flaw that makes the implant prone to loosening, especially when routine force is applied to the area.
Cartiva Implant Shrinking
The Institute has also theorized that Cartiva implants may shrink after they are implanted due to the lack of hydration. Dr. Baravarian from the Institute stated in an article published in Podiatry Today, that the Cartiva implant is largely water-based and is soaked in a water medium prior to being implanted.
Experts theorize that after Cartiva is implanted it may lack hydration from the surrounding joint, allowing it to shrink and slip into the bone.
Bone Weakening Around The Implant
The University Foot & Ankle Institute has also suggested the Cartiva implant may fail due to the bone surrounding the implant being too weak, and failing to provide adequate support. The smooth surfaces of the implant combined with a lack of support to hold the implant in place could allow loosening and implant slipping.
Cartiva Implant Failure Symptoms
Some of the most common symptoms of Cartiva failure include;
- Swelling and Inflammation
- Redness or tenderness at the joint site
- Infection
- Feeling of instability when standing or walking
- Discomfort in the affected joint
- Nerve damage
- Post operative pain
- Reduced range of motion in the toe
- Cysts
Cartiva FAQs
Who manufactures Cartiva?
The Cartiva synthetic cartilage implant was originally manufactured by Cartiva, Inc. in 2015 and was sold to Wright Medical Group in 2018. Wright Medical Group was then purchased by Stryker in 2020. Cartiva Lawsuits are being filed against all manufacturers involved in the process of designing and manufacturing the big toe implant. Lawsuits are not being pursued against foot surgeons who implanted Cartiva, unless there is an independent basis for a medical malpractice claim.
How long will a Cartiva implant last?
While Cartiva has been hailed as a revolutionary treatment that is designed to last a lifetime, evolving medical research has shown many recipients experience Cartiva complications within several months to two years after receiving the implant.
Allegations Raised in Cartiva Implant Lawsuits
Individual injury claims and Cartiva class action lawsuits are being pursued for individuals who had received a defective and unreasonably dangerous Cartiva implant and required a revision or replacement surgery. Allegations being raised against the manufacturer in Cartiva toe implant lawsuits include;
- Designing and selling a defective and unreasonably dangerous toe implant;
- Failure to adequately research the life expectancy of Cartiva implants;
- Failure to accurately validate the wear of Cartiva toe implants;
- Failure to properly investigate post-market reports of Cartiva problems;
- Failure to promptly issue a Cartiva implant recall;
- Putting profits before the safety of patients.
Cartiva Implant Failure Lawsuit Settlements
As a result of the manufacturer’s apparent faulty design of the Cartiva SCI decision and desire to profit before the health and safety of patients, Cartiva lawsuit settlements may be available.
The product liability lawyers at Saiontz & Kirk, P.A. are actively investigating Cartiva lawsuits for individuals who have suffered complications resulting in the need for revision or replacement surgery.
How Much Is A Cartiva Settlement Worth?
The value of potential Cartiva settlements could vary depending on several factors. Individuals who suffered injuries resulting in permanent disability may be entitled to higher settlement payouts than those with successful revision surgery.
In determining the amount of any individual Cartiva lawsuit payout, the following are some of the common factors that will be taken into consideration when negotiating or evaluating any offers:
- How many Cartiva implants an individual received;
- The specific injuries caused by Cartiva;
- The amount of any past or future medical expenses;
- The amount of any lost wages or loss of earning capacity;
- The level of impact Cartiva has caused on the individual’s overall physical and mental health or well-being;
- The past and future amount of pain and mental anguish suffered by the individual;
Are there any costs to hire a Cartiva toe implant lawyer?
There are absolutely no out-of-pocket costs to hire our Cartiva lawyers for your toe implant failure case. Potential claims are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis. Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm for their Cartiva lawsuit — regardless of their individual financial resources.
You pay nothing up front to hire our Cartiva implant lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturer. Our law firm receives nothing unless we win your case!
What are the steps in a Cartiva case evaluation?
Complete Our Case Evaluation Request Form. Providing contact information and some information about your Cartiva implant problems.
Get Contacted by Saiontz & Kirk, P.A. You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.

