Why Is There A Port Catheter Lawsuit?
Individuals throughout the United States are actively pursuing port catheter lawsuits against certain manufacturers, due to dangerous design defects associated with a number of different implantable port catheter devices.
Totally implantable vascular access devices (TIVADs), often called port catheters or port a caths, are frequently used for venous access during long-term therapy, such as chemotherapy ports and dialysis ports. However, there is increasing evidence that certain designs may pose a much greater risk of TIVAD problems, which have not been adequately disclosed to patients or the medical community.
The TIVAD lawyers at Saiontz & Kirk provide free consultations and claim evaluations to help determine whether financial compensation may be available. There are never any out-of-pocket expenses to hire our attorneys, and we receive no fees or expenses unless we win!
Who Qualifies For A Port Catheter Lawsuit?
As a result of the manufacturers’ failure to adequately test their medical devices and warn the medical community, our lawyers are actively pursuing TIVAD lawsuit settlements for individuals who have experienced any of the following injuries caused by a defectively designed implantable venous access system;
- Perforations of tissues, vessels and organs
- Sepsis
- Catheter-Related Bloodstream Infections
- Fluid Leak Injuries
- Deep Vein Thrombosis (DVT)
- Hemorrhaging or Bleeding Injuries
- Fluid buildup on the heart
- Irregular Heartbeat
- Severe and persistent pain
- Organ Failure or Dysfunction
- Wrongful death
- Other injuries caused by fractured PowerPort catheter
Info About Port Catheter Lawsuits On This Page
Which Port Catheters Are Named In Lawsuits?
Port Catheter Lawsuits: Product Liability or Medical Malpractice?
How Long Do I Have To File A Port Catheter Lawsuit?
How Much Is A Port Catheter Lawsuit Worth?
What is A Port Catheter/Port-a-Cath?
What Are The Problems With Port a Cath?
Port-a-Cath Injuries & Side Effects
Port Catheter Recalls Over Defects & Problems
How Do You Know If Something Is Wrong With Your Port?
Which Port Catheters Are Named In Lawsuits?
Multiple different products have been named in port catheter lawsuits filed in recent years, each raising allegations of design defects and indicating that the medical device manufacturer placed profits over consumer safety, by concealing the risks and failing to adequately warn patients or the medical community about potential TIVAD problems.
Bard PowerPort Lawsuits
Most of the U.S. litigation involves Bard PowerPort lawsuits filed against Becton Dickinson and its Bard subsidiaries, alleging that the widely used implantable port has an unreasonably dangerous design, which may increase the risk of a fracture, fissure or crack in the port catheter. This may result in serious injuries, blood clots, infection and even wrongful death.
Lawsuits over the Bard PowerPort catheter fractures claim that the manufacturers knew about the PowerPort fracture and migration risks for years, but failed to fully disclose the extent of the problems. The development of cracks or fissures in the material may also result in bacteria that leads to a higher risk of Bard PowerPort infection problems. However, instead of altering the design or issuing a port catheter recall, it appears that Bard placed profits before patient safety.
Our product liability lawyers are investigating PowerPort fracture, migration and infection lawsuits for individuals who received any of the following Bard TIVAD models;
- Bard PowerPort
- Bard Powerflow™
- Bard SlimPort
- Bard X‑Port™
- Bard MRI Ports
- Bard Groshong Catheter
- Bard Titanium Port
- Bard Vaccess CT
Angiodynamics Smart Port Lawsuits
AngioDynamics, Inc. has faced Smart Port lawsuits in recent years, raising allegations that the catheters used in the Smart Port implantable venous access devices are prone to fissuring, cracking, fracturing, and infections due to manufacturing defects.
Similar to PowerPort lawsuits, plaintiffs claim that the catheters in the Smart Port devices contain high concentrations of barium sulfate particles, which leads to the silicone being too thick and improper mixing of the particles. The AngioDynamics port lawsuits allege that the manufacturer could have prevented these problems with design changes to the SmartPort venous access system, which may have avoided catheter failure events and serious injuries.
Our product liability lawyers are investigating AngioDynamics fracture, migration and infection lawsuits for individuals who received any of the following Angiodynamics TIVAD models;
- Angiodynamics/Navilyst BioFlo Port
- Angiodynamics/Navilyst SmartPort
- Angiodynamics/Navilyst SmartPort CT
- Angiodynamics/Navilyst Vaxcel
- Angiodynamics/Navilyst Vortex
- Angiodynamics/Navilyst Xcela
Smiths Medical Port-a-Cath Lawsuits
Smith Medical is another manufacturer that has faced Port-A-Cath lawsuits brought by individuals who received this widely known implanted port device. Lawsuits over port-a-caths also allege that the product has design defects, which make them prone to fracturing and migrating through an individual’s body.
Our product liability lawyers are investigating Smiths Medical TIVAD fracture, migration and infection lawsuits for individuals who received any of the following models;
- Smiths Deltec
- Smiths Medical P.A.S. Port
- Smiths Medical Port-a-Cath
- Smiths Medical Power P.A.C.
Port Catheter Lawsuits: Product Liability or Medical Malpractice?
In addition to pursuing port catheter product liability lawsuits against manufacturers of defective devices, some claims have resulted in TIVAD medical malpractice lawsuits against the surgeons who implanted the port. However, these situations are very rare, and typically involve complications that arise immediately after the TIVAD is implanted.
In most cases, the evidence establishes that the doctor followed the proper standard of care and complications were the result of design defects or inadequate warnings provided by the manufacturer. Saiontz & Kirk will NOT pursue a port catheter lawsuit against your surgeon unless you specifically authorize us to do so and there is clear evidence of medical malpractice.
Port Catheter Product Liability Lawsuits
Product liability lawsuits against the device manufacturer can provide the full financial compensation individuals deserve for injuries experienced, either through a port catheter settlement or verdict at trial. These claims allow multiple users to pool their resources to prove that the implantable TIVAD was defective, and then establish that this defect resulted in their injury. There are generally three types of defects that can be used to establish a port catheter product liability lawsuit:
- Design defects: These are inherent flaws in the design of the product itself. To prove a design defect, users typically demonstrate that there was a safer, feasible, and practical alternative design that the manufacturer could have used.
- Manufacturing defects: These occur when something goes wrong in the production process that makes the product unsafe. Establishing a manufacturing defect in a port catheter claim is more rare, and typically requires a showing that the product deviated from its intended design and that the deviation is what caused it to be unsafe.
- Failure to Warn defects: These involve inadequate instructions, failure to provide sufficient warnings about potential risks, or misleading marketing material provided to either consumers or the medical community. To prove a marketing defect, plaintiffs show that the manufacturer knew or should have known about a potential risk, that they failed to adequately disclose that risk, and that an injury resulted.
Port Catheter Medical Malpractice Lawsuits
A medical malpractice lawsuit over port catheters requires a plaintiff to establish that a specific healthcare professional was negligent and breached the standard of care, causing harm to a patient. To establish that complications with an implantable port a cath constitute a medical malpractice lawsuit depends on several factors:
- Breach of Duty: If your healthcare provider did not follow accepted professional standards in installing, maintaining, or monitoring your implantable port, and that failure led to your injury, this could constitute a breach of duty.
- Causation: It must be demonstrated that your injury is a direct result of the healthcare provider’s breach of duty. That is, would this injury have occurred if the healthcare provider had followed the proper standard of care?
- Harm: You must have suffered harm or injury as a result of this breach of duty. This can include physical harm, mental distress, additional medical costs, or lost earnings.
How Long Do I Have To File A Port Catheter Lawsuit?
The timeframe within which you must file a lawsuit for an injury related to a TIVAD—or any medical device, for that matter—is determined by what’s known as the “statute of limitations.” The exact length of time varies by jurisdiction and the specifics of the case. However, in most cases it usually ranges from 2 to 3 years from the date of the injury or from the time when you discovered or should have discovered the injury.
For instance, in some states, the statute of limitations for personal injury cases is 2 years, but the clock may not start ticking until the harm or injury has been discovered, a concept known as the “discovery rule.” This is often applied in port catheter lawsuits, where it is not immediately apparent that the complications were caused by a defective product.
Determining the specific statute of limitations for a port catheter lawsuit requires an analysis of the circumstance in each case, and a review of the applicable laws in the state where the device was implanted or failed. The lawyers at Saiontz & Kirk provide free consultations and claim evaluations to help determine how long you may have to file a port catheter lawsuit.
How Much Is A Port Catheter Lawsuit Worth?
The value of each individual implantable port catheter injury lawsuit will vary widely, depending on a number of factors. Each case is unique, so the average port catheter lawsuit payout or settlement will include some claims with very high values, and others with lower values. An attorney can help you understand the potential value of your case based on the specifics of your injury and the laws in your jurisdiction.
The amount of a TIVAD lawsuit settlement may be determined on the following factors;
- Severity of the Injury
- Medical Expenses
- Physical Pain and Suffering
- Punitive Damages
- Lost Wages and Earning Capacity
- Loss of enjoyment of life
- Funeral expenses
Allegations Raised in Port Catheter Lawsuits
Product liability lawsuits over defective implantable port catheter designs are being filed against multiple manufacturers. The TIVAD lawsuits allege that the manufacturers:
- Knew about fracture, migration and infection risks for years, and failed to adequately investigate the cause of the problems;
- Failed to warn physicians and consumers that the implantable port catheters may weaken and fracture, causing fragments to migrate throughout the body;
- Negligently continued to sell dangerous and defective implantable ports, while concealing information from consumers, regulators and the medical community;
- Failed to alter the design of the devices to ensure the safety of patients.
- Failed to issue a recall and placed desire for profits before consumer safety.
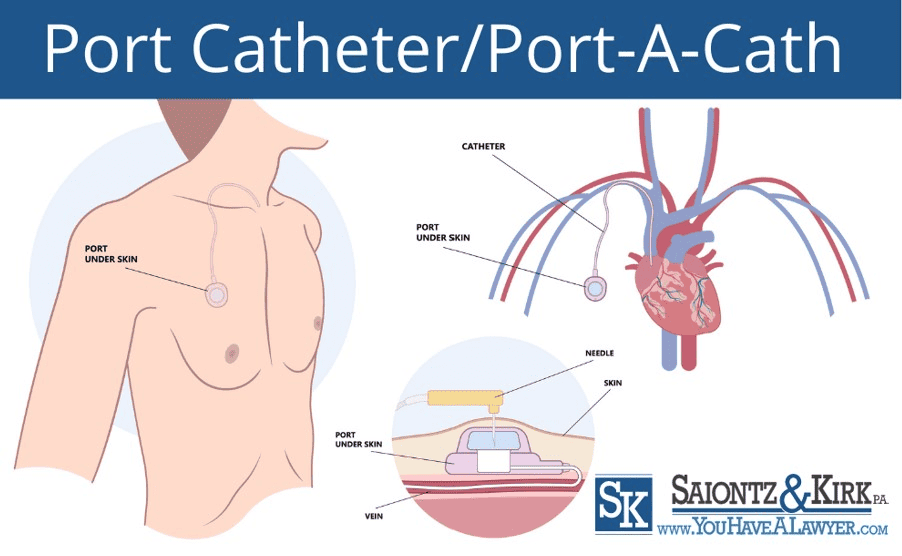
What is A Port Catheter/Port-a-Cath?
A port catheter, also known as a Port-a-Cath, is a totally implantable venous access devices (TIVAD) used to provide easy and long-term access to a patient’s veins for various medical treatments. Port catheters are known by many names including: IV ports, chemo ports, subcutaneous implanted port and totally implantable venous access devices (TIVADs).
Port catheters consist of two main components: the portal and the catheter:
- The portal, typically made from a self-sealing silicone material, is surgically inserted beneath the patient’s skin. It appears as a small bump and can be punctured repeatedly with a needle without compromising its integrity.
- The catheter, a slender plastic tube, is attached to the portal and is threaded into a central vein such as the jugular vein, subclavian vein, or the superior vena cava. This positioning allows for easy administration of medications, fluids, blood products, or nutrients directly into the bloodstream.
What Are Implanted Ports Used For?
Implanted ports are used in various medical conditions and treatments that require long-term intravenous therapy or frequent access to veins. Some of the most common medical conditions and treatments that may require the use of implantable venous access port includes:
- Chemotherapy: Patients undergoing chemotherapy often require frequent administration of medications directly into their bloodstream. Implantable chemo ports provide a reliable and convenient access point for delivering chemotherapy drugs.
- Antibiotic Therapy: Certain infections may require long-term antibiotic therapy, and implantable ports can be used to administer antibiotics directly into the bloodstream over an extended period.
- Nutrition: Patients who are unable to consume food orally or have malabsorption issues may require parenteral nutrition, which involves providing nutrients directly into the veins. Implantable ports facilitate the administration of parenteral nutrition.
- Hemodialysis: In some cases, implantable ports can be used for hemodialysis, a procedure that filters waste products from the blood in individuals with kidney failure. However, other types of vascular access, such as arteriovenous fistulas or grafts, are more commonly used for this purpose.
- Blood Transfusions: Implantable ports can be used to administer blood transfusions when necessary. This allows for easier and more efficient transfusion procedures.
- Frequent Blood Draws: Some medical conditions or treatments require frequent blood tests for monitoring purposes. Implantable ports can simplify the process by providing an easily accessible point for blood collection.
Are port catheters safe?
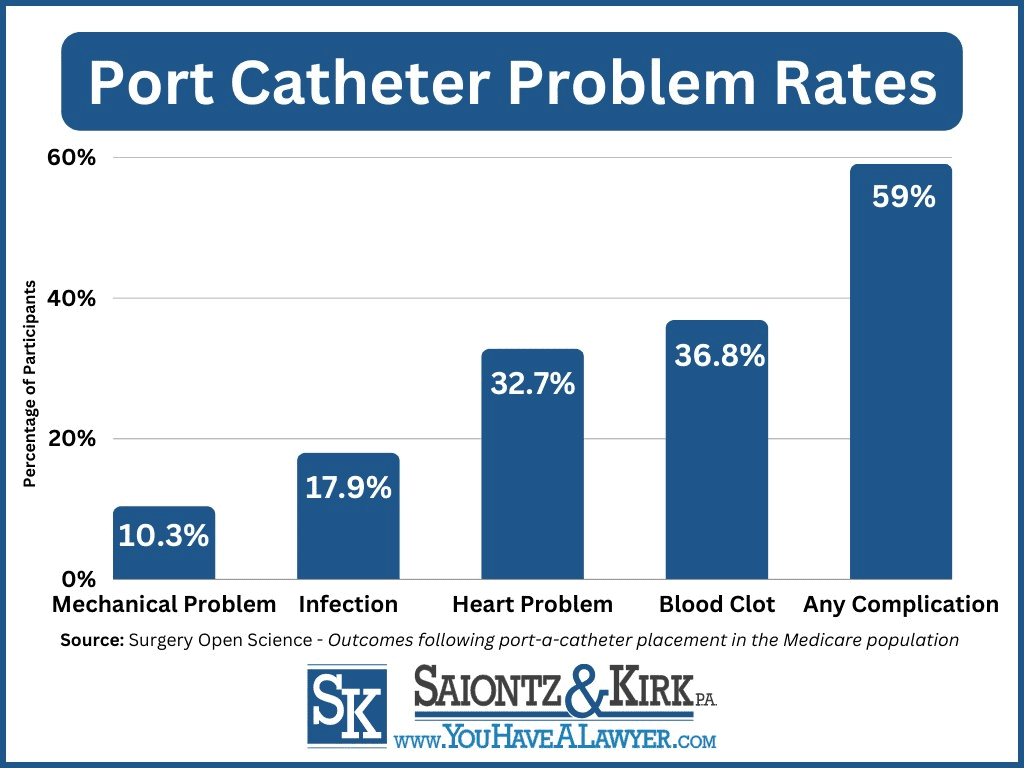
While implantable port catheters are generally considered safe, a 2021 study published in Surgery Open Science revealed that out of 93,756 patients who received a port-a-catheter between 2007 and 2012, an alarming 59.04% experienced complications within just five years of implantation.
The most common injuries caused by implantable port catheter included;
- Any complication– 55,353 (59.0%)
- Blood clots in blood vessels– 34,499 (36.8%)
- Heart problems– 30,625 (32.7%)
- Infection– 16,745 (17.9%)
- Mechanical complication– 9,670 (10.3%)
What are the types of implanted ports?
The types of implanted port devices can be classified according to several different aspects:
- Single Lumen: This port features a single access point, or septum, which makes it the most commonly used type.
- Double Lumen: This type features two access points to a vein, typically in a triangular shape. This is beneficial when multiple medications need to be administered simultaneously.
- Power-Injectable Ports: Both single- and double-lumen ports can be power-injectable. This means that healthcare providers can inject contrast dyes into these ports for performing imaging scans such as CT scans and MRIs.
What Are The Problems With Port a Cath?
The safety of port catheters has come under scrutiny in recent years, due to an alarming number of associated problems. The FDA has taken action to issue multiple port catheter recalls over design defects that may cause the devices to crack, fracture, and migrate within the body.
Moreover, certain port catheter devices have been known to cause infections and leaks, leading to the improper delivery of medications and fluids into a patient’s bloodstream.
Common Complications of Port Placement
While serious complications with port catheters can occur at any point in time during their use, studies have shown individuals are at most risk of infections after receiving an implantable port device. The risk of infection is a significant concern after implantation and may arise due to bacterial contamination during the procedure or inadequate sterile techniques.
In most situations, an infection after a port catheter placement does not result in a product liability lawsuit. However, infections that develop longer after the port is implanted may be the result of design defects.
Bleeding can also occur during or after the port implantation procedure due to mispositioning or misplacement which causes damage to blood vessels or inadequate hemostasis. Hematomas or excessive bleeding can lead to pain, swelling, and compromised catheter function.
Port Catheter Fracture Risks
A TIVAD catheter fracture can cause serious injuries or death to individuals when they break or migrate in the body. This can occur if the catheter was defectively designed, or when the catheter becomes less durable due to constant flexing.
The resulting fragments can scatter throughout the circulatory system which could increase the risk of a port catheter fatality from;
- Blood clots in arteries
- Cardiac punctures
- Cardiac arrythmia
- Severe hemorrhage
Recently, a number of Bard PowerPort lawsuits have been filed by individuals claiming the PowerPort contains a design defect which makes the device prone to fracturing and migrating. Learn more about Bard PowerPort lawsuits here.
Design defects that may cause catheter fractures
A port catheter design defect refers to a flaw or issue in the structural makeup of the device that can lead to severe consequences for patients. Some of the most common design defects associated with an implantable port catheter failure have included;
- Defective Catheter Material and Flexibility: The material and flexibility of the catheter can contribute to fracture risk. If the catheter material is rigid or lacks flexibility, it may be more susceptible to stress and fracture. Design defects in the catheter material or inadequate flexibility can compromise its structural integrity and increase the likelihood of fracture.
- Catheter Pinch-Off Events: A common mechanism of catheter fracture is constant compression between the first rib and clavicle, known as the pinch-off syndrome. This occurs when the catheter is compressed or squeezed between these bony structures due to the design or positioning of the port system. The continuous pressure on the catheter can weaken it over time, leading to fracture.
- Catheter Fixation: Improper catheter fixation within the port chamber can create stress points or tension, making the catheter more prone to fracture. If the design does not provide adequate support or fixation mechanisms to secure the catheter properly, it can result in catheter movement and potential fracture.
- Catheter Length and Position: The length and positioning of the catheter within the body can also influence the risk of fracture. If the catheter is excessively long or placed in a manner that causes it to bend or twist, it may be more susceptible to stress and subsequent fracture. Proper design considerations for catheter length and positioning are important to minimize the risk of fracture.
Port Catheter Migration Risks
Catheter Migration is a rare but potentially life-threatening complication in which the catheter may become dislodged from the port and move to other vital organs in the circulatory system, such as the heart. Oftentimes, these incidents require immediate surgical intervention and extraction to prevent fatal injuries.
Why does a port catheter move?
The two most common reasons for a port catheter to move are;
- Surgical Mistakes: A surgical error by medical professionals such as excessive force, tension, or accidental trauma during catheter insertion or manipulation can result in catheter breakage. If the catheter breaks, it may migrate to different locations within the body.
- Manufacturing defects: Several implantable catheters that have been linked to migration risks in recent years have been found to contain defective designs that allow catheters to leak or develop cracks that can break away and travel through the bloodstream.
Pulmonary embolism from catheter migration
Catheter migration not only poses a cardiovascular risk to patients but also increases the likelihood of developing a pulmonary embolism. With this condition, blood clots block the flow of blood in the lungs’ arteries, reducing or even completely cutting off the blood supply. This can result in several serious consequences, including;
- Restricted blood flow
- Reduced oxygen levels in the blood
- Damage to the lungs and other organs caused by insufficient oxygenation
Port Catheter Infection Risks
Unlike traditional catheters, TIVADs stay in place for longer durations and require maintenance which involves cleaning before and after use. However, even with proper care, it has been discovered that the designs of certain port catheters can harbor bacteria and viruses, causing Catheter-related bloodstream infections (CRBSI). These infections often occur due to the following reasons;
- Breach in sterile technique: A breach in sterile technique during catheter insertion or use can result in bacteria access in the bloodstream, which can lead to severe bloodstream infections and sepsis.
- Catheter Fractures: One of the main reasons for the development of these infections is the presence of holes and cracks in the catheter material, which allow bacteria and other pathogens to seep through. The issue with these tiny openings is that they are often too small for the body’s immune cells to traverse, leaving the pathogen to wreak havoc and potentially leading to severe infections that can be difficult to treat.
Some of the most common infections caused by a port catheter include;
- Coagulase-negative staphylococci
- Candida
- Escherichia coli
- Fungal infections
- Klebsiella
- Staphylococcus aureus
Port Catheter Leaks
Implantable port catheter leaks occur when there is a break or damage in the catheter, allowing fluids to escape into the body. This can be dangerous because it can affect medication delivery, cause infection, and even lead to severe complications such as blood clots or tissue death. Patients who experience a leak may also suffer from pain, swelling, and discomfort at the site of the catheter.
Catheter leaks can occur due to various device failures or surgical errors, including;
- Catheter damage
- Catheter disconnection
- Catheter blockage
- Port chamber failures
- Faulty valves
- Injection port failures
- Defective locking mechanisms
- Broken or improper sealings
Port-a-Cath Injuries & Side Effects
A defectively designed or improperly placed TIVAD can increase the risk of serious side effects for individuals. In conjunction with the delay of critical and potentially life saving treatments could advance the stages of disease such as cancer, the side effects of a defective implantable port catheter may also cause;
- Infections (sepsis or septic shock)
- Deep Vein Thrombosis (DVT)
- Internal bleeding
- Fluid buildup on the heart
- Irregular heartbeat
- Leaking medications into the body
- Chronic and persistent pain
- Perforations of tissues, vessels
- Organ damage
- Emergency surgery
- Wrongful death
Port Catheter Recalls Over Defects & Problems
There have been multiple TIVAD recalls initiated by medical device manufacturers and the U.S. Food & Drug Administration (FDA) in recent years, as well as lawsuits filed by affected patients alleging device failure and related health complications.
Specific examples of recalls include devices like the Bard PowerPort, PORTACATH, and PORTACATH II, with the recalls prompted by complications such as catheter misplacement, fracture, and malfunction, which may lead to serious medical conditions.
Bard PowerPort duo M.R.I. Implantable Port Recall
On March 25, 2021, a Bard PowerPort recall was issued impacting PowerPort duo M.R.I. Implantable Ports. The Bard PowerPort device recall was prompted by several reports of issues during usage, such as difficulty in flushing, infusion, and/or aspiration, and incidents of septum dislodgments.
Bard Access Systems, Inc., a fully owned branch of Becton, Dickinson and Company (BD), clarified in the recall notice that a dislodged septum might allow fluid to seep into the patient’s implanted port. This could potentially lead to pain and injury, depending on how quickly the leak is detected and the volume and nature of the leaking fluid.
If an irritant or vesicant solution were to leak from the PowerPort, it could cause significant toxic and necrotic damage, necessitating immediate medical action to treat the affected tissue. Additionally, if there is difficulty or an inability to flush, infuse, or aspirate, it could delay necessary treatment, warrant further medical intervention, and potentially result in the need to abandon the port and proceed with surgical removal or replacement.
SmartPort Recall
AngioDynamics, Inc. issued a SmartPort recall on February 22, 2021, over an increased risk of infections to patients, warning certain Implantable Infusion Port kits and Valved introducers may have been compromised and not sterile for use.
Bard PowerPort Implantable Port Catheter Recall
Bard Peripheral Vascular Inc issued a Bard PowerPort recall for certain implantable ports on October 01, 2019, warning there is a risk of an incorrectly attached tunneler with a barb tip for a 6Fr catheter instead of the intended barb tip for a 9.6Fr catheter. The manufacturer cautioned this mistake can cause significant delays and complications during surgery.
Port-A-Cath Recall
On September 10, 2013, Smiths Medical initiated a port-a-cath recall that included its PORT-A-CATH and PORT-A-CATH II Implantable Venous and Arterial Access Systems and Introducer Sets. The recall of the port a cath devices was prompted by reports of possible delamination of the catheter tip, a condition that could potentially lead to embolism, vascular occlusion, or other severe health-related implications.
Vortex MP Port Recall
The recall of the Vortex MP Port, initiated on September 20, 2022, was prompted by a packaging and labeling issue. The product label failed to include a crucial statement regarding the catheter’s construction material.
Specifically, it omitted the fact that the silicone catheter contained DEHP, a chemical known for its potential health risks. This omission presented significant concerns because patients and healthcare providers rely on accurate and comprehensive labeling to make informed decisions regarding the use of medical devices.
How Do You Know If Something Is Wrong With Your TIVAD Port?
Depending on what is wrong with your implantable port, you may experience a number of symptoms that could include;
- Pain or Discomfort: If you have discomfort, tenderness, or pain around the area of your port, especially when it’s being used, it might be a sign of a problem.
- Redness or Swelling: Any redness, swelling, or warmth over the skin where the port is located could be signs of an infection.
- Fever: Fever or chills might also be an indication of an infection associated with the port.
- Fluid Leakage: If there’s any leakage of fluid from the area of the port, it’s a sign that it could be malfunctioning.
- Difficulty using the port: If your healthcare provider has difficulty flushing or withdrawing blood from the port, there might be a blockage or the catheter might have moved out of place.
- Changes in the skin around the port: Any changes in the skin around the port, such as rashes, discoloration, or unusual marks could be a sign of an underlying issue.
If you suspect that there are problems with your port catheter, seek immediate medical attention.
Are There Any Costs to Hire a Port Catheter Lawyer
There are absolutely no out-of-pocket costs to review your case or hire our attorneys. Potential claims are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis.
Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm for their port catheter lawsuit — regardless of their individual financial resources.
You pay nothing up front to hire our lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturers. Our law firm receives nothing unless we win your case!
What are the steps in a Port Catheter case evaluation?
Complete Our Case Evaluation Request Form. Providing contact information and some information about your catheter injury case.
Get Contacted by Saiontz & Kirk You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.

