Is There A Tepezza Hearing Loss Lawsuit?
Yes. The product liability lawyers at Saiontz & Kirk are investigating Tepezza hearing loss lawsuits against Horizon Therapeutics Inc. for former users who may be entitled to settlement benefits, due to the manufacturer’s failure to adequately warn about the potential side effects.
Tepezza (teprotumumab-trbw) was approved by the FDA in January 2020, as the first drug for treatment of treat thyroid eye disease, which is caused by hyperthyroidism and linked to Graves’ disease. Although the drug maker was aware of Tepezza hearing problems during clinical trials, inadequate warnings were provided for users and the medical community.
Over the first few years the drug was on the market, users experienced Tepezza hearing loss side effects at an alarming rate, resulting in problems like:
- Sensorineural Hearing loss
- Ringing in the ears (Tinnitus)
- Difficulty hearing
- Ear plugging sensation
- Clicking and popping sounds
- Muffled sounds
- Deafness
- Sensitivity to certain sounds
- Problems hearing words in noisy environments
- Own voice sounding abnormally loud and low-pitched
- Pain mimicking an ear infection
- Dizziness and balance problems
- Needing to turn up the volume of the television or radio
If hearing loss warnings and instructions about the importance of monitoring for hearing problems on Tepezza had been provided, users may have been able to avoid permanent and disabling injuries.
Saiontz & Kirk is pursuing Tepezza side effects lawsuits for individuals who have received any number of Tepezza infusions and have been diagnosed with any of the following conditions;
- Permanent hearing loss
- Partial hearing loss
- Tinnitus (ringing of the ears)
The medication lawyers at Saiontz & Kirk provide free consultations and case evaluations to help determine if Tepezza lawsuit settlement benefits may be available for you or a loved one who suffered hearing loss or Tinnitus.
Is There a Tepezza Class Action Lawsuit?
There is a not a Tepezza class action lawsuit being pursued on behalf of all former users who received the infusion treatment. However, the U.S. Judicial Panel on Multidistrict Litigation has ordered all Tepezza hearing loss claims filed throughout the federal court system to be consolidated for pretrial proceedings in a Tepezza multidistrict litigation (MDL).
Under the Tepezza MDL, U.S. District Judge Thomas Durkin in the Northern District of Illinois will coordinate discovery into common issues that apply to all claims, and likely schedule a series of early test trials to help promote Tepezza settlement negotiations.
Tepezza Lawsuit Status Update
- With the size of the Tepezza litigation growing as more individuals learn about the undisclosed hearing risks, a Tepezza MDL (multidistrict litigation) was established in June 2023, centralizing the lawsuits before U.S. District Judge Thomas Durkin in the Northern District of Illinois for coordinated discovery and pretrial proceedings.
- Tepezza hearing loss lawsuits are now being pursued by individuals nationwide who have suffered permanent hearing loss after receiving Tepezza infusions for the treatment of thyroid eye disease and Grave’s disease.
- Studies and case reports have found that side effects of Tepezza increase the risk of hearing loss or Tinnitus.
- Since the infusion was introduced in 2020, cases of permanent hearing damage from Tepezza have been reported by users nationwide.
2026 Tepezza Lawsuit Update
March 2024 Update: Plaintiffs have selected four Tepezza hearing loss claims to be prepped for early trial dates. On March 1, 2024, plaintiffs submitted a list of bellwether selections, which includes two lawsuits involving individuals who suffered permanent hearing loss from Tepezza, and the other two include both permanent hearing loss and tinnitus injuries caused by the drug.
November 2023 Update: U.S. District Judge Thomas Durkin issued a case management order on November 1, 2023, instructing the parties to begin selecting claims that will be part of the bellwether discovery pool in early 2024.
July 2023 Update: As Tepezza hearing loss lawsuits continue to be filed, the FDA has updated the prescribing information guide for Tepezza, warning of the potential risk of severe and permanent hearing loss. The new warnings urge doctors to carefully consider the benefits and risks of treatment and to evaluate patients for symptoms of hearing loss before, during, and after treatment with Tepezza.
June 2023 Update: A Tepezza multidistrict litigation has been formed after the U.S. Judicial Panel on Multidistrict Litigation (JPML) issued a transfer order on June 2, 2023, ordering all Tepezza lawsuits filed throughout the federal court system to be consolidated before U.S. District Judge Thomas Durkin in the Northern District of Illinois for coordinated pretrial proceedings.
May 2023 Update: In response to a motion to consolidate a growing number of Tepezza lawsuits raising similar allegations that Horizon Therapeutics failed to warn about hearing loss risks, the U.S. Judicial Panel on Multidistrict Litigation (JPML) is scheduled to hear oral arguments on May 25, 2023, to determine whether to centralize the claims into one Tepezza MDL (Multidistrict Litigation).
January 2023 Update: At least 10 separate Tepezza lawsuits claim Horizon Therapeutics aggressively marketed the drug for the treatment of thyroid eye disease while failing to disclose the risks of permanent hearing loss and other hearing injuries.
November 2022 Update: As of November 2022, no Tepezza recall has been issued, nor has the manufacturer updated the Tepezza warning label to add new information about hearing problems or the importance of monitoring for hearing loss during treatment.
October 2022 Update: Amid increasing reports of Tepezza side effects, individuals who have been left with hearing problems are now actively filing Tepezza lawsuits, alleging the manufacturer downplayed the risk of permanent hearing impairment and other hearing-related injuries.
May 2021 Update: A study was published in the Journal of the Endocrine Society finding 65% of patients receiving Tepezza injections experienced partial or permanent hearing loss, ringing of the ears or other hearing related injuries.
2020 Update: Tepezza was approved by the FDA in January 2020, as the first drug for treatment of treat thyroid eye disease (TED). However, since the infusion entered the market, cases of permanent hearing damage from Tepezza have been reported by users nationwide.
Tepezza Lawsuit Information On This Page
Tepezza Hearing Problems
How Does Tepezza Cause Hearing Loss?
Studies Linking Tepezza and Hearing Loss
Tepezza Hearing Loss Settlement Information
Tepezza Hearing Problems
The link between Tepezza and hearing problems should have been clear to the drug maker in the very early stages of its development and throughout the FDA approval process. Clinical trial data suggested that users were experiencing hearing loss and other hearing-related problems, but strong warnings were not provided for patients or doctors.
In the clinical trials, 10% of patients treated with Tepezza reported new hearing impairment problems, compared to zero new hearing problems reported among individuals in the placebo group. Some of the hearing impairments reported during clinical trials included;
- Deafness: Deafness is defined as a partial or complete loss of hearing. Depending on the severity of the hearing loss, deafness can range from mild to profound. People who are deaf may use a variety of communication methods, including sign language, lip reading, and written language.
- Eustachian tube dysfunction: The Eustachian tube is a small passageway that connects the middle ear to the back of the throat and serves to equalize pressure in the middle ear and drain fluid from the ear. Eustachian tube dysfunction is when the tube is not able to open and close properly. This can lead to a feeling of fullness or pressure in the ear, pain, tinnitus, and hearing loss.
- Hyperacusis: Hyperacusis is a hearing disorder that makes it difficult for people to tolerate everyday sounds. Hyperacusis can be caused by damage to the inner ear or by problems with the auditory nerve. In some cases, people with hyperacusis may also experience tinnitus, or ringing in the ears.
- Hypoacusis: Hypoacusis, also known as partial deafness, is a condition in which a person has difficulty hearing certain sounds. Symptoms of hypoacusis may include difficulty hearing conversation or television, ringing in the ears, or feeling like one’s ears are “full.”
- Autophony: Autophony is the experience of hearing one’s own voice differently from how other people hear it. This can include hearing your voice echoing, sounding muffled, or being louder than usual. In some cases, autophony may be experienced as a symptom of tinnitus.
While Horizon Therapeutics clinical trials only indicated a 10% risk of hearing problems from Tepezza, a 2021 study published by The Endocrine Society found as many as 65% of Tepezza recipients were experiencing hearing loss or other new hearing problems.

Despite clinical data, independent studies and numerous case reports linking Tepezza to permanent hearing damage risks early on, Horizon did not issue an updated Tepezza drug label warning until July 2023.
In addition to the alert regarding potential irreversible hearing loss, the FDA approved update also directs healthcare professionals to be vigilant in monitoring their patients for auditory issues prior to treatment, throughout the course of treatment, and in the follow-up period.
Who is the Tepezza Lawsuit Against?
Tepezza lawsuits are being filed against Horizon Therapeutics Inc., which is a wholly owned subsidiary of Horizon Therapeutics PLC.
Tepezza was granted Orphan Drug designation when approved, which allowed the medication to be introduced without extensive testing, since there is no other drug treatment available for thyroid eye disease. However, Tepezza clinical trials have been criticized since the studies only involved a total of 84 patients, which may not have accurately portrayed all of the potential side effects or their severity.
Even though the FDA granted approval under the special designation, drug makers still have a duty to ensure the safety of the medication they are selling, and to issue warning labels about the potential side effects.
According to allegations raised in Tepezza lawsuits, Horizon reported to the FDA that the hearing loss side effects from Tepezza were only temporary, suggesting that the problems would resolve after months of stopping infusion treatments. However, medical journals have published case studies documenting no improvements in hearing loss in the months after receiving Tepezza injections.
How Does Tepezza Cause Hearing Loss?
It appears that Tepezza causes hearing loss through the same mechanism of action used to treat thyroid eye disease.
Tepezza works by inhibiting the insulin-like growth factor-1 and the insulin-like growth factor-1 receptor pathway. It is known that insulin-like growth factor-1 protects inner ear hair cells from noise-induced damage, ischemia and medication toxicity. Therefore, the drug makers knew or should have known that inhibiting this pathway may lead to Tepezza-induced hearing loss.
Despite these known risks, Horizon Therapeutics failed to adequately warn that Tepezza may cause hearing loss, or instruct patients and doctors about the importance of regular audiological testing to detect hearing problems on Tepezza.
Studies Linking Tepezza and Hearing Loss
In addition to clinical trials identifying hearing problems from Tepezza, multiple case reports and peer reviewed medical journal studies published after the infusion was on the market in the United States have found a potential link between Tepezza and hearing loss.
Tepezza Linked to 65% Increased Risk of Hearing Problems
The findings of a study published in the Journal of the Endocrine Society were presented at a virtual annual meeting of The Endocrine Society in March 2021, indicating that 65% of patients who took Tepezza reported hearing problems, such as ringing of the ears, autophony, a sensation that their ears were plugged, or total hearing loss.
Stanford researchers evaluated the side effects of Tepezza in 26 patients who received at least four infusions. Seventeen of the patients, or 65%, reported developing hearing problems after Tepezza injections. In addition, 23% reported suffering hearing loss; 27% developed tinnitus, a ringing in the ears; and 12% reported ear plugging sensation side effects. Another 29% said they developed autophony, where their own voices sounded unusually loud to themselves.
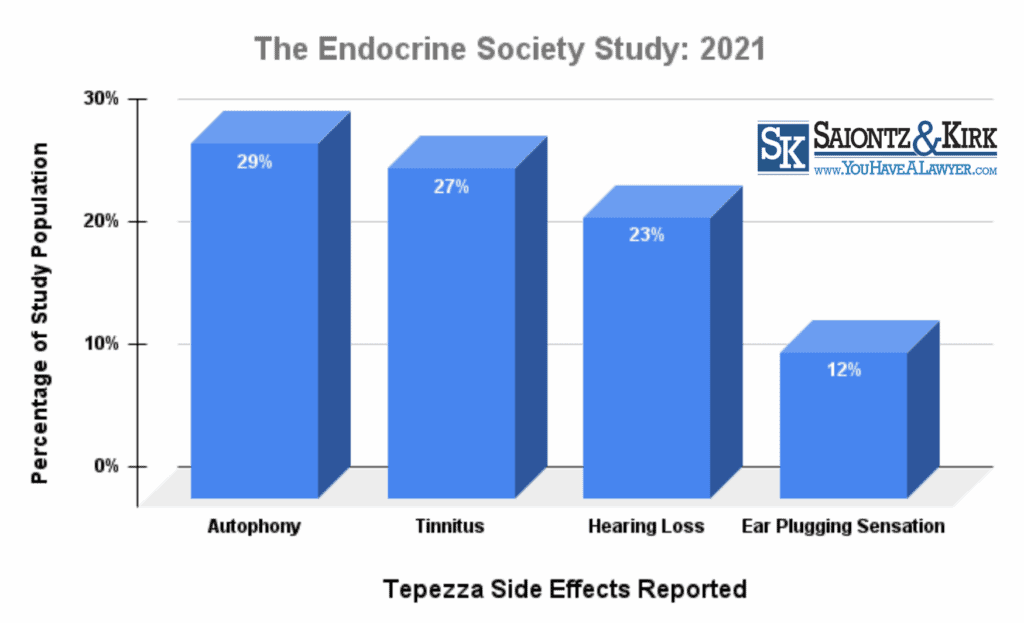
Patients reported experiencing Tepezza side effects after an average of 3.6 infusions. While some patients saw Tepezza-related hearing problems lessen or disappear after several months, researchers indicated the follow up period was only three months, and was too short to properly assess whether the problems were truly reversible.
The study authors indicated that doctors need to be made more aware of the potential hearing loss risks caused by Tepezza, and recommended all new patients be screened before receiving the infusion and to conduct baseline audiogram testing.
Hearing Problems Reported During Tepezza Clinical Trials
Prior to the FDA granting Tepezza’s approval through a fast-tracked Orphan Drug designation, Horizon submitted the drug for FDA approval based on limited clinical trial data involving less than 100 patients.
In the clinical trial data submitted to the FDA, Horizon even reported that 10% of individuals treated with Tepezza suffered new hearing problems. However Horizon classified the Tepezza hearing problems as only temporary, indicating they would resolve after months of stopping the injections, which has not been the case with many Tepezza patients who have been left with permanent hearing loss and hearing problems.
Tepezza Post-Marketing Safety Analysis
According to the results of a 19 month post-marketing safety analysis of hearing problems caused by Tepezza was released in February 2022, reporting similar findings that upward of 10% of those receiving Tepezza injections experienced hearing problems. The Safety analysis reported;
“in this 19-month analysis (Jan. 2020 – Aug. 2021), approximately 10% of all cases reported to the safety database have included a hearing-related event. The most frequently reported hearing event was hypoacusis (reduction in hearing), followed by tinnitus (ringing in the ears).”
Tepezza Hearing Loss Case Reports
In a January 2022 study published in the medical journal Ophthalmic Plastic and Reconstructive Surgery, researchers reviewed four specific cases of Tepezza hearing loss, stating;
“Teprotumumab may cause a spectrum of potentially irreversible hearing loss ranging from mild to severe”
Researchers further warned;
“Due to the novelty of teprotumumab and the lack of a comprehensive understanding of its effect on hearing, the authors endorse prospective investigations of hearing loss in the setting of teprotumumab treatment.“
As a result of their findings, researchers recommended prescribing doctors adopt surveillance protocols including the use of audiogram and tympanometry before, during and after Tepezza infusions to monitor new hearing impairment symptoms.
Permanent Hearing Loss After Tepezza Use: Case Report
Researchers published the findings of a case report in The Annals of Otology, Rhinology, and Laryngology in August 2022, indicating that a 61 year old female with Graves ophthalmopathy suffered hearing loss, sound distortion, and tinnitus following Tepezza treatments.
Despite stopping Tepezza for four months, researchers stated her hearing loss and tinnitus symptoms remained unchanged. Researchers stated;
“This is one of the first descriptive cases of ototoxicity resulting in irreversible sensorineural hearing loss in the setting of treatment with teprotumumab.”
Allegations Raised in Tepezza Lawsuits
The lawyers at Saiontz & Kirk are reviewing Tepezza class action lawsuits and individual claims for individuals diagnosed with hearing loss or tinnitus. According to allegations raised in Tepezza lawsuits, Horizon Therapeutics has:
- Failed to properly research the side effects of Tepezza before introducing the drug;
- Knew or should have known that Tepezza may cause permanent hearing loss;
- Knew or should have known that Tepezza may cause Tinnitus;
- Failed to warn about the potential hearing problems from Tepezza;
- Failed to promptly issue Tepezza recalls or safety warnings for users and the medical community.
Tepezza Hearing Loss Lawsuit Settlements
As a result of Horizon Therapeutics’ apparent decision to place a desire for profits before the health and safety of patients, settlement benefits may be available through a Tepezza hearing loss lawsuit.
Since the litigation remains in the early stages, with the first Tepezza lawsuits filed in late 2022, there have not been any global Tepezza settlements or lawsuit payouts. However, as individuals begin to learn about the allegations raised against Horizon over the manufacturer’s alleged failure to warn, it is anticipated that additional Tepezza lawsuits will be filed throughout 2023. As the litigation grows, the manufacturer may engage in Tepezza hearing loss settlement talks to resolve claims.
How Much Is A Tepezza Settlement Worth?
The value of potential Tepezza hearing loss lawsuit could vary depending on the specific severity of hearing problems experienced. Individuals who suffered permanent hearing loss or tinnitus will be entitled to substantially higher settlement payouts than those with partial hearing loss or minor hearing damage.
In determining the amount of any individual Tepezza hearing loss lawsuit payout, the following are some of the common factors that will be taken into consideration when negotiating or evaluating any offers:
- How many Tepezza infusions an individual received;
- The specific hearing problems caused by Tepezza;
- Whether or not the hearing problems from Tepezza are permanent;
- The amount of any past or future medical expenses incurred as a result of Tepezza injuries;
- The amount of any lost wages or loss of earning capacity;
- The level of impact Tepezza has caused on the individual’s overall physical and mental health or well-being;
- The past and future amount of pain and mental anguish suffered by the individual;
How Long Will It Take To Receive A Tepezza Lawsuit Payout?
Since Horizon Therapeutics is still reviewing post-marketing safety analysis data, and has yet to issue a Tepezza recall or updated Tepezza warnings, it is expected that this litigation may take several years for individuals who suffer hearing loss injuries to receive fair and appropriate Tepezza settlement offers.
As the litigation moves forward, it is expected that a small group of “bellwether” cases will be selected by the court, involving facts that are representative of larger numbers of Tepezza hearing loss claims. Those bellwether cases will go through case-specific discovery and a series of early trials, to help the parties gauge how juries are likely to respond to certain evidence that will be repeated throughout the Tepezza lawsuits.
As Tepezza trial dates approach, or following a number of Tepezza lawsuit payouts ordered by juries, our lawyers expect that Horizon Therapeutics will consider engaging in settlement negotiations to avoid each individual lawsuit being set for trial in courts throughout the U.S.
Are there any costs to hire a Tepezza lawyer?
There are absolutely no out-of-pocket costs to hire our Tepezza lawyers for your hearing loss case. Potential claims are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis. Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm for their Tepezza lawsuit — regardless of their individual financial resources.
You pay nothing up front to hire our Tepezza hearing loss lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the drug manufacturer. Our law firm receives nothing unless we win your case!
What are the steps in a Tepezza case evaluation?
Complete Our Case Evaluation Request Form.Providing contact information and some information about your Tepezza side effects.
Get Contacted by Saiontz & Kirk You will be contacted by our law firm to help determine if financial compensation may be available through a Tepezza hearing damage lawsuit.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to file a Tepezza lawsuit.

