Is There an Ozempic Lawsuit?
Yes. Individuals across the United States are pursuing Ozempic lawsuits against the drug manufacturer, which claim the drug manufacturer failed to adequately warn users about severe gastrointestinal issues such as stomach paralysis and intestinal obstruction, as well as an increased risk of nonarteritic anterior ischemic optic neuropathy (NAION), a serious eye condition.
A growing body of medical research and adverse events reported to the U.S. Food and Drug Administration (FDA) have identified a potential link between gastroparesis and Ozempic, as well as other side effects that were not adequately disclosed to users or the medical community.
According to allegations raised in gastroparesis lawsuits being pursued against Novo Nordisk and its subsidiaries, the drug makers placed a desire for profits before consumer safety by failing to properly research its diabetes and weight loss drugs, and failing to warn about the risk of severe and long-lasting stomach problems and vision problems users may experience.
The product liability lawyers at Saiontz & Kirk provide free consultations and claim evaluations for individuals diagnosed with gastrointestinal or vision problems after taking Ozempic, Wegovy and other semaglutide drugs.
Is there an Ozempic Class Action Lawsuit?
The U.S. Judicial Panel on Multidistrict Litigation issued a transfer order on February 2, 2024, instructing all GLP-1 stomach paralysis lawsuits involving Ozempic, Wegovy, Mounjaro and other similar GLP-1 drugs to be centralized in the U.S. District Court for the Eastern District of Pennsylvania before U.S. District Judge Gene E. K. Pratter for coordinated pretrial proceedings.
Which Semaglutide Drugs Will Be Named In Lawsuits?
Lawsuits are being pursued for users of any diabetes or weight loss drugs containing the active ingredient semaglutide. In addition to claims for users of Ozempic, the same allegations are being pursued in Wegovy lawsuits and Rybelsus lawsuits, which have also been linked to the same gastric and vision problems.
Our lawyers are also investigating potential Mounjaro lawsuits for individuals who experienced gastrointestinal injury after taking this similar medication, which contains the active ingredient tirzepatide.
Who Qualifies for an Ozempic Lawsuit?
Financial compensation may be available for individuals who received Ozempic or Wegovy injections, or took Rybelsus tablets, and suffered any of the following complications:
- Gastroparesis (Stomach paralysis)
- Ileus (Intestinal obstruction)
- Gastric Stasis
- Pancreatic Cancer
- Deep Vein Thrombosis
- Pulmonary Aspiration
- Vision Changes/Blindness
- Other gastrointestinal problems
To help determine the Ozempic settlement amount that you or a loved one may be eligible to receive, request a no-obligation case evaluation today.
2026 Ozempic Lawsuit Update
Following recent studies and case reports linking Ozempic and gastrointestinal problems, individuals are now seeking compensation through an Ozempic lawsuit over the manufacturer’s failure to warn about the side effects of the GLP-1 receptor agonist drugs.
- March 3, 2026 Update: The federal court overseeing GLP-1 drug lawsuits continues organizing pretrial proceedings for claims alleging Ozempic, Wegovy and Mounjaro caused stomach paralysis and other severe gastrointestinal injuries. Judge Karen Marston is expected to move the litigation toward the next stage of coordinated discovery and begin identifying representative cases that could eventually be prepared for bellwether trial testing.
- January 1, 2026 Update: As the new year begins, the federal GLP-1 multidistrict litigation remains focused on allegations that popular diabetes and weight-loss medications caused gastroparesis, stomach paralysis and other serious digestive injuries. The court signaled that resolving scientific disputes over whether the drugs can cause long-term gastrointestinal damage will be a key issue guiding discovery and pretrial proceedings throughout 2026.
-
More Ozempic Lawsuit Update
November 3, 2025 Update: The federal judge presiding over the GLP-1 MDL granted additional time for both sides to address several major legal and scientific disputes that could influence the direction of the litigation. The issues include whether federal law preempts failure-to-warn claims and whether existing evidence sufficiently links GLP-1 medications to gastroparesis and other gastrointestinal injuries. The revised schedule delays preparations for the first bellwether trials until at least the following summer.
September 15, 2025 Update: The number of claims in the GLP-1 gastroparesis litigation continues to rise, with more than 2,400 cases pending in federal court before U.S. District Judge Karen Marston. The MDL is advancing through coordinated discovery as attorneys prepare the groundwork for early bellwether trials that could help gauge how juries respond to evidence linking the drugs to severe digestive injuries.
March 28, 2025 Update: Plaintiffs in the GLP-1 drug litigation formally opposed efforts by drug manufacturers to dismiss most claims in the consolidated master complaint. Attorneys representing injured users argued that the lawsuits involve more than simple failure-to-warn allegations, asserting that evidence may support claims involving negligent misrepresentation, design defects and breach of warranty related to the safety of the medications.
February 5, 2025 Update: Eli Lilly and Novo Nordisk have requested Judge Marston to dismiss most of the claims in the master complaint, except for the Failure to Warn allegations. The motion specifically aims to eliminate claims related to breaches of warranty, fraudulent and negligent misrepresentation, unfair trade practices, design defects, negligence, and demands for medical monitoring, asserting that the plaintiffs’ claims are invalid on grounds that the plaintiffs have not sufficiently demonstrated the legal elements necessary for each claim.
October 2, 2025 Update: According to the latest report from the U.S. Judicial Panel on Multidistrict Litigation, the federal GLP-1 MDL has grown to 2,809 pending lawsuits involving Ozempic, Wegovy, Mounjaro and similar medications. Plaintiffs across the country claim the drugs caused severe gastrointestinal complications, including stomach paralysis, chronic vomiting and intestinal obstruction.
March 28, 2025 Update: Plaintiffs in the GLP-1 drug litigation formally opposed efforts by drug manufacturers to dismiss most claims in the consolidated master complaint. Attorneys representing injured users argued that the lawsuits involve more than simple failure-to-warn allegations, asserting that evidence may support claims involving negligent misrepresentation, design defects and breach of warranty related to the safety of the medications.
January 1, 2025 Update: Judge Marston issued a scheduling order for the Ozempic litigation, requiring drugmakers to file motions to dismiss by January 24, 2025, arguing federal preemption of failure-to-warn claims. Plaintiffs must respond by March 18, with replies due by April 14, after which oral arguments will be scheduled. Fact discovery on general causation must be completed by July 2, 2025, with expert reports exchanged in July and August. Monthly status conferences will begin January 14, and a decision on preemption may come by year-end.
July 2024 Update: A new study has found that Ozempic and Wegovy users face an almost 8-fold increased risk of developing vision loss side effects , particularly from a condition called non-arteritic anterior ischemic optic neuropathy (NAION), which can cause cause sudden changes in vision and may lead to blindness.
June 2024 Update: The latest docket report from the Judicial Panel on Multidistrict Litigation (JPML) indicates that there are currently 101 lawsuits filed against the manufacturers of Ozempic and other GLP-1 agonist drugs in the Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation.
May 2024 Update: Judge Pratter has issued a case management order indicating that on June 14, 2024, a dedicated Science Day will be held in the Ozempic litigation. During this session, involved parties will present scientific findings in both support and opposition to the claims asserting that the use of Ozempic has resulted in significant gastrointestinal issues, such as gastroparesis, ileus, and intestinal obstructions, among other adverse conditions, experienced by the plaintiffs.
February 2024 Update: The JPML issued a transfer order (PDF) on February 2, 2024, ordering all lawsuits filed over Ozempic, Wegovy, Mounjaro and other GLP-1 stomach paralysis side effects to be centralized in the U.S. District Court for the Eastern District of Pennsylvania as part of a Glucagon-Like Peptide-1 Receptor Agonist Products Liability Litigation.
January 2024 Update: The U.S. JPML will hold a hearing on January 25, 2024, at the Central District of California Courthouse to discuss the consolidation of all GLP-1 stomach paralysis lawsuits, including those involving Ozempic, Wegovy, Mounjaro, and similar cases, to assess if centralizing these suits is appropriate.
December 2023 Update: A group of plaintiffs have filed a motion to transfer (PDF) with the U.S. Judicial Panel on Multidistrict Litigation requesting that all lawsuits filed over gastroparesis side effects caused by Ozempic, Wegovy, Mounjaro and other similar Type II diabetes or weight loss drugs be consolidated for pretrial proceedings.
October 2023 Update: A publication in the Journal of the American Medical Association (JAMA) warned about stomach paralysis risks of Ozempic and other GLP-1 agonist medications commonly prescribed for weight management. The study indicated that Ozempic triples the risk of gastroparesis, or stomach paralysis, when compared to other therapeutic alternatives.
September 2023 Update: The U.S. Food and Drug Administration (FDA) released an Ozempic warning label update on September 22, 2023, stating that the gastrointestinal disorder, Ileus, is a possible side effect of semaglutide drugs used for weight loss and type II diabetes management.
August 2023 Update: The first of what is expected to be many Ozempic and Mounjaro lawsuits was filed over gastroparesis side effects in the U.S. District Court for the District of Louisiana on August 2, 2023.
July 2023 Update: A CNN investigative report cautioned that a rising number of people are experiencing stomach paralysis after taking Ozempic and Wegovy. The report also emphasized the mounting evidence suggesting that the manufacturers of these diet drugs knew about the potential side effects.
June 2023 Update: The American Society of Anesthesiologists (ASA) recommended that users temporarily discontinue Ozempic, Wegovy before elective surgery due to the risk of experiencing nausea and vomiting that could cause suffocation during anesthesia procedures.
Ozempic Lawsuit Settlements
As the Ozempic litigation remains in the early stages, there have not been any global Ozempic lawsuit settlements to date. However, our lawyers expect that as additional medical research is presented and potentially thousands of Ozempic gastrointestinal injury lawsuits are filed by users throughout the United States, the manufacturers could decide to settle the claims to avoid a lengthy litigation.
How much is an Ozempic settlement payout worth?
The compensation you could recover in an Ozempic gastroparesis lawsuit will vary depending on the extent of the injuries sustained. The impact of gastroparesis from Ozempic can result in very different consequences, with some users requiring prolonged hospitalization and being left with permanent disabilities. The individual circumstances in each case will be considered in negotiating any Ozempic settlement.
How is an Ozempic lawsuit settlement calculated?
Similar to other pharmaceutical drug and product liability lawsuits resulting in personal injuries to users, multiple factors will be considered when established an Ozempic lawsuit settlement amount, including;
- Extent of the Injury: The gravity and enduring nature of the gastrointestinal injury experienced by the plaintiff will play a pivotal role in shaping the compensation amount. Low or mild discomfort would likely result in a lesser settlement figure compared to a persistent injury or disability.
- Medical Expenses: Costs related to the injury, such as hospitalization, prescribed medications, surgeries, and follow-up treatments, will be a significant determinant in the settlement amount.
- Future Medical Needs: Predicted medical expenses, especially if the injury might require ongoing treatments, will also be evaluated when considering a settlement payout.
- Lost Wages: If the injury resulted in the plaintiff missing work or becoming unable to work either temporarily or permanently, these lost earnings will be accounted for in the settlement calculation..
- Pain and Suffering: This encompasses both the physical pain and emotional turmoil undergone by the individual due to the injury.
- Strength of Evidence: How compelling and credible the evidence is in establishing a link between Ozempic and the gastrointestinal injuries will be paramount. The science is continuing to evolve and strengthen, and if the manufacturer determines a jury is more likely to believe Ozempic caused gastroparesis, it will drive larger settlement amounts.
To find out how much your potential Ozempic lawsuit settlement could be worth, contact our lawyers for a free consultation to discuss the specifics of your case.
Problems With Ozempic, Wegovy and Rybelsus
Semaglutide is the active ingredient in Ozempic, Wegovy and Rybelsus. The prescription medication was formulated by Novo Nordisk and first received FDA approval for use as a type 2 diabetes treatment in December 5, 2017, resulting in the introduction of Ozempic as a once-weekly injectable GLP-1 receptor agonist designed to regulate blood sugar by enhancing insulin production.
Shortly after the drug was released, it was aggressively marketed by Novo Nordisk and quickly became popular for its ability to help with weight loss, which was considered an “off-label” use.
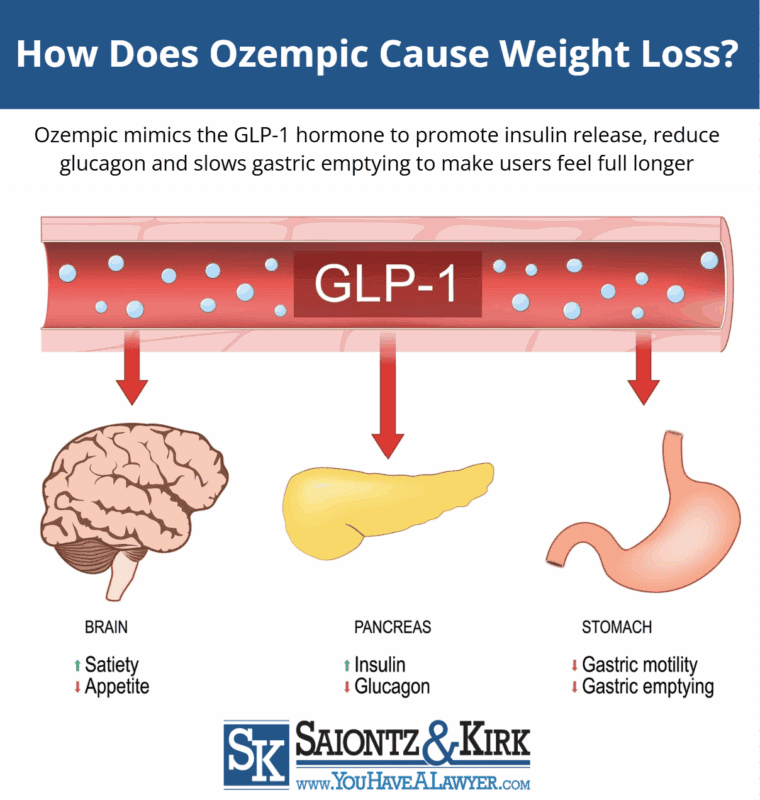
Ozempic has been shown to help some lose weight by its ability to mimic the GLP-1 hormone that is released from the intestines after eating. This causes insulin release, slows gastric emptying, and suppresses secretion of glucagon (a hormone that increases blood sugar). However, this same mechanism of action can leave users with painful stomach problems from Ozempic.
By mimicking GLP-1’s action, Ozempic can slow the movement of food through the stomach, which may help users feel full longer, reduces appetite and leads to caloric excretion. The drug maker failed to warn that for some users, Ozempic delayed gastric emptying can cause permanent damage to the stomach and result in gastroparalysis, or stomach paralysis.
With Ozempic quickly becoming a top selling drug due to off label prescriptions as an obesity medicine, the manufacturer subsequently released an oral version of semaglutide in a tablet for the treatment of Type 2 diabetes named Rybelsus, which was approved by the FDA in 2019.
While the drug maker has made claims that it released the diabetes medications in a tablet form to accommodate those who cannot tolerate Ozempic injections, many critics have claimed the Rybelsus tablet was released in an effort to make the drug more affordable and boost appeal to the masses seeking semaglutide as an obesity treatment.
Recognizing the demand for the diabetes drug Ozempic among individuals seeking off label weight loss benefits, Novo Nordisk introduced another variant of semaglutide called Wegovy. In June 2021, the FDA approved Wegovy as a weight loss medication for adults battling obesity and those overweight due to specific health issues.
Ozempic’s success for obesity treatment prompted other drug makers to formulate their own version of GLP-1 receptor agonists. By May 2022, Eli Lilly had developed and received approval for its drug tirzepatide, which was branded as Mounjaro.
However, as millions of individuals have turned to semaglutide and tirzepatide for weight reduction, a surge of adverse event reports involving users developing severe gastrointestinal issues after taking Ozempic, Wegovy, Rybelsus and Mounjaro have been reported.
Ozempic Side Effects Reported to the FDA
Lawsuits now allege that Novo Nordisk put profits before customer safety, by ignoring a shocking number of side effects that were reported to the FDA by individuals taking Ozempic, Wegovy and Rybelsus.
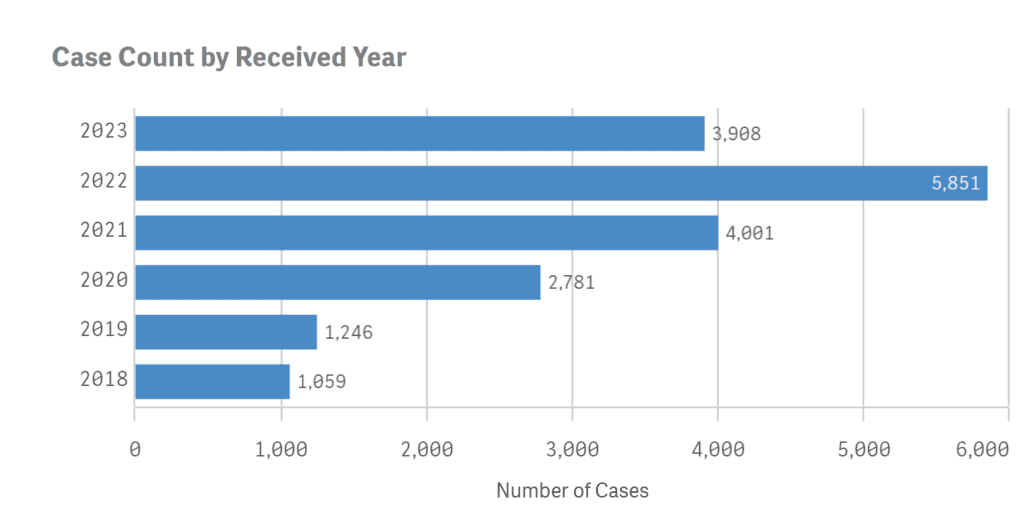
According to the FDA’s FAERS database, the number of adverse reactions each year since Ozempic, Rybelsus and Wegovy were released have climbed significantly, reaching a total of 18,846 adverse events as of June 30, 2023.
The number of Ozempic side effects reported per year are as follows;
- 2018 Adverse Events: 1,059
- 2019 Adverse Events: 1,246
- 2020 Adverse Events: 2,781
- 2021 Adverse Events: 4,001
- 2022 Adverse Events: 5,851
- 2023 Adverse Events: (As of June 30, 2023) 3,908
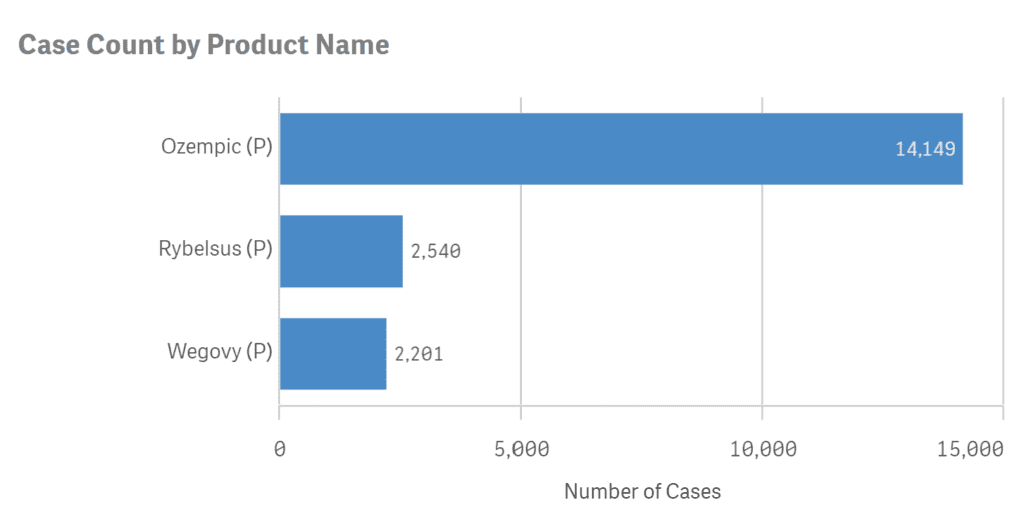
According to the FDA’s database, the number of adverse events reported for each semaglutide drug since they have been released up until June 30, 2023, are as follows;
- Ozempic adverse events: 14,149
- Rybelsus adverse events: 2,540
- Wegovy adverse events: 2,201
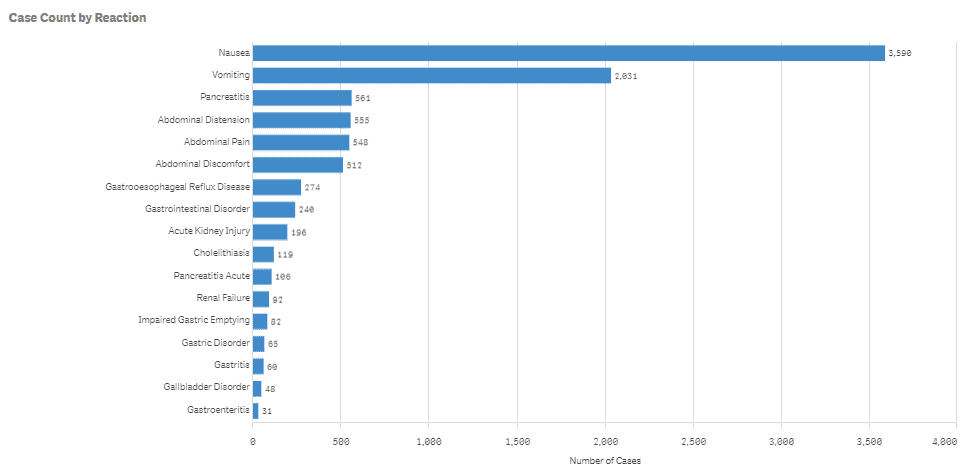
Many of the Ozempic side effects reported to the FDA have included gastrointestinal problems, including;
- Nausea: 3,590
- Severe Vomiting: 2,031
- Pancreatitis: 607
- Abdominal Distension: 555
- Abdominal Pain: 548
- Abdominal/Stomach Pain: 512
- Gastroesophageal Reflux Disease: 274
- Gastrointestinal Disorder: 240
- Acute Kidney Injury: 196
- Cholelithiasis: 119
- Renal Failure: 92
- Impaired Gastric Emptying: 82
- Gastric Disorder: 65
- Gastritis: 60
- Gallbladder Disorder: 48
- Gastroenteritis: 31
Ozempic Warning Label Failures
Despite thousands of adverse reactions being reported by individuals taking Ozempic within the first several years of being on the market, the manufacturer failed to issue a timely warning.
Rather than further investigating the nearly 20,000 adverse events reported to the FDA, Novo Nordisk pumped billions into advertising and celebrity endorsers speaking out on the weight loss benefits, producing billions of dollars in revenue while users continue to report new digestive illnesses.
2023 Ozempic Warning Over Ileus Gastrointestinal Risks
In response to an increasing number of reports regarding gastrointestinal issues, the U.S. Food and Drug Administration (FDA) announced a semaglutide warning label update on September 22, 2023, warning that Ozempic’s warning label would be updated to include certain gastrointestinal side effects on September 22, 2023.
Specifically, the newly modified label now features a warning for Ozempic ileus gastrointestinal side effects.
Symptoms of ileus induced by Ozempic bear a resemblance to gastroparesis symptoms that have been reported by users to the FDA in recent years. Although ileus and gastroparesis are distinct conditions, they have overlapping symptoms. These can include nausea, vomiting of undigested food, abdominal pain, bloating, severe dehydration, a sense of fullness after only a few bites, solidification of undigested food within the stomach, acid reflux, fluctuating blood sugar readings, reduced appetite, weight loss, undernutrition, and a diminished overall quality of life.
Ozempic and Gastroparesis
Individuals are now filing lawsuits against the makers of Ozempic claiming that the manufacturer was well aware of reports of gastric disorders and symptoms that align with the onset of severe gastroparesis, yet failed to further investigate the severity or permanency of the injury risks.
Stomach Paralysis From Ozempic
Gastroparesis arises when the stomach muscles malfunction, inhibiting their ability to adequately process and move food. This impedes the stomach from efficiently emptying itself.
Ozempic is intentionally designed to slow gastric emptying to aid in weight loss. As a result, lawsuits claim the manufacturer failed to warn that the continuous use of Ozempic and Wegovy can cause some individuals to experience prolonged and chronic delays in stomach emptying.
Symptoms of Gastroparesis from Ozempic may include;
- Nausea
- Vomiting
- Feeling full quickly when eating
- Abdominal bloating
- Stomach pain

Ozempic & Wegovy Clinical Trials Show Symptoms of Gastroparesis
Lawsuits allege the drug maker was aware of the risk of stomach paralysis injuries during clinical trials, yet failed to update warning labels or adequately warn users and the medical community.
During Ozempic clinical trials, 20% of participants experienced nausea, and 10% noted occurrences of vomiting, which are both symptoms prevalent in cases of gastroparesis.
During Wegovy clinical trials, close to 44% of Wegovy users experienced nausea and nearly 25% of the test subjects also reported incidents of vomiting.
The heightened occurrences of nausea and vomiting incidents found in the Wegovy trial align with the theory that semaglutide causes an increased risk of gastroparesis, since Wegovy is more than double the dose at 2.4mg compared to Ozempic’s 1mg dose. However, individuals using Ozempic for weight loss typically receive the higher doses of semaglutide similar to what is contained in Wegovy.
Ozempic Triples Stomach Paralysis Risks Among Users
With millions of Americans opting for injectable weight management medications like Ozempic and Wegovy, a study published in the Journal of the American Medical Association (JAMA) on October 5, 2023, revealed that Ozempic users are three times more likely to experience stomach paralysis than individuals using alternative oral weight loss medications.
The study assessed the side effects of semaglutide and liraglutide injectable GLP-1 agonist drugs, and compared the rate of similar side effects to those taking Contrave (bupropion-naltrexone), an oral medication for weight management. Within the study group, 4,144 liraglutide users and 612 semaglutide users reported gastrointestinal issues, compared to 654 among Contrave users.
According to the findings, Ozempic was associated with the highest rate of new stomach paralysis cases, followed by liraglutide and then Contrave.
- 1% – Ozempic
- 0.7% – liraglutide
- 0.3% – Contrave
Further, the research suggests that injectable semaglutide and liraglutide drugs are four times more likely to cause bowel obstruction and nine time more likely to cause pancreatitis when compared to Contrave.
Ozempic Delays Gastric Emptying Warning
In July 2023, the American Society of Anesthesiologists (ASA) spotlighted the potential hazards linked to a condition called delayed gastric emptying from Ozempic and Wegovy, particularly for users undergoing surgical procedures. Due to side effects like nausea and vomiting from Ozempic, patients might face heightened risks of choking or inhaling food into the respiratory system while under general anesthesia or deep sedation.
This caution regarding Wegovy and Ozempic was part of broader ASA recommendations, issued in light of the medications’ increasing popularity as weight management solutions. The guidelines suggest daily users of drugs like Ozempic or Wegovy refrain from taking them on the day of surgery. Those on a weekly regimen should discontinue their use one week prior to the surgical procedure. It’s also recommended that patients liaise with an endocrinologist during this medication pause to ensure effective diabetes management.
Furthermore, the guidelines advise medical professionals to think about postponing surgeries if the patient reports significant gastrointestinal symptoms on the day of the procedure, such as pronounced nausea, vomiting, abdominal discomfort, or bloating.
Ozempic, Wegovy and Rybelsus Linked to Vomiting Risks
In a study published in the medical journal, The BMJ, on January 29, 2024, researchers identified a significant link between the use of semaglutide-based drugs, such as Ozempic and Wegovy, and an elevated risk of adverse gastrointestinal side effects, including nausea, vomiting, and diarrhea.
This systematic review, conducted by researchers from the Beijing University of Chinese Medicine and the University of Chicago, analyzed data from 76 studies to assess the safety and efficacy of fifteen glucagon-like peptide-1 (GLP-1) receptor agonists used in the treatment of type 2 diabetes.
The findings demonstrate that, although GLP-1 agonists can be effective in lowering haemoglobin A1c and fasting plasma glucose levels, higher doses of these medications significantly increase the risk of gastrointestinal issues. Compared to placebo groups, the use of semaglutide drugs were associated with a notably higher incidence of;
- Nausea: 246% increased risk,
- Vomiting 325% increased risk,
- Diarrhea 137% increased risk, and
- Drug discontinuation due to these adverse events :161% increased risk.
Ozempic Gallbladder Injuries and Side Effects
Research indicates a connection between Ozempic and severe gastrointestinal events, specifically gallstones (cholelithiasis) and gallbladder inflammation (cholecystitis). While our lawyers are not actively pursuing Ozempic gallbladder lawsuits, studies that identified this risk also should have resulted in stronger warnings about the risk of more severe and long-lasting gastroparesis problems.
In 2017, the journal Diabetes, Obesity & Metabolism published a pivotal meta-analysis examining the effects of Ozempic on pancreatitis and gallbladder disease. This research was the first to link Ozempic with an increased risk of gallbladder diseases including gallstones and gallbladder inflammation due to delayed gastric emptying.
When the gallbladder doesn’t regularly or fully empty, bile can accumulate, potentially raising the risk of gallstone formation. These gallstones can then obstruct the bile ducts, leading to inflammation or infection, possibly necessitating gallbladder removal.
Typically, gallbladder disease symptoms manifest as intense pain in the upper right or center of the abdomen. This pain might be constant or come and go, and it can be paired with intense stomach discomfort, nausea, vomiting, and fever.
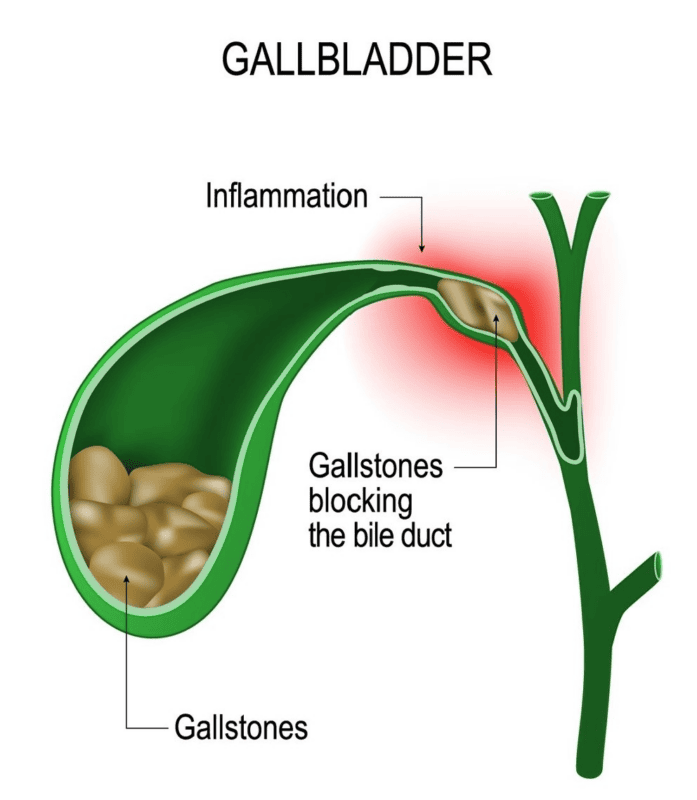
In March 2022, Novo Nordisk provided updated Ozempic gallbladder warnings about the risk of gallbladder disease, but failed to add information about the risk of gastroparesis or stomach paralysis from Ozempic.
At this time, our lawyers are not reviewing Ozempic gallbladder injury cases.
Ozempic Vision Loss Side Effects – NAION
In a study published in the medical journal JAMA Ophthalmology on July 3, 2024, researchers indicate Ozempic and Wegovy users may be seven times more likely to experience vision changes, including blurred vision or vision loss, when compared to those given other medications.
Dr. Jimena Tatiana Hathaway and her research team explored the link between Ozempic and increased risks of nonarteritic anterior ischemic optic neuropathy (NAION), particularly in individuals with type 2 diabetes or who are overweight or obese.
Non-arteritic anterior ischemic optic neuropathy (NAION) is a condition characterized by a sudden decrease in vision due to reduced blood flow to the optic nerve. This lack of blood supply can cause permanent damage to the nerve, leading to partial or complete loss of vision in the affected eye.
The study involved data from 16,827 patients evaluated by neuro-ophthalmologists, and focused on patients with type 2 diabetes or those who were overweight or obese, comparing those prescribed semaglutide to those using non-glucagon-like peptide-1 receptor agonist (GLP-1 RA) medications.
The findings indicate that over a 36 month period, Ozempic or Wegovy users faced significantly higher risks of developing NAION compared to those using non-semaglutide medications, indicating;
- Type II Diabetes Patients: NAION risks increased from 1.8% in non-semaglutide users to 8.9% in semaglutide users, representing a 710% increased absolute risk.
- Overweight or Obese Patients: NAION risks increased from 0.8% in non-semaglutide users to 6.7% among semaglutide users, representing a 590% increased absolute risk.

Allegations Raised In Ozempic Lawsuits
Individuals throughout the United States are currently pursuing Ozempic lawsuits against Novo Nordisk and its subsidiaries raising allegations that the manufacturer,
- Failed to adequately research the link between semaglutide and vision problems and gastrointestinal problems
- Failed to warn about the increased risk of gastrointestinal side effects and NAION
- Falsely advertised the medications as a safe weight loss drug.
- Failed to issue an Ozempic recall to ensure consumers were aware of the health risks.
Are there any costs to hire an Ozempic Lawyer?
There are absolutely no out-of-pocket costs to review your case or hire our attorneys. Ozempic lawsuits are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis.
Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm for their Ozempic, Wegovy or Rybelsus lawsuit — regardless of their individual financial resources.
You pay nothing up front to hire our Ozempic lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturer. Our law firm receives nothing unless we win your case!
How to File an Ozempic Lawsuit
Complete Our Case Evaluation Request Form. Providing contact information and some information about your Ozempic problems.
Get Contacted by Saiontz & Kirk You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.

