Paragard Copper IUD Lawyers Review Cases Nationwide
The Paragard IUD is a small T-shaped plastic device wrapped in copper wires, which many women have implanted in their uterus as a form of long-acting birth control. The copper IUD is designed to prevent pregnancy for up to 10 years, but women are told that it is completely reversible, allowing them to have children in the future.
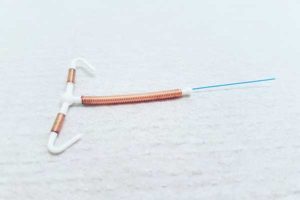
Due to an unreasonably dangerous and defective design, the Paragard IUD is prone to fracture and break during removal procedures, leaving women with painful and permanent injuries.
The product liability lawyers at Saiontz & Kirk are reviewing potential Paragard IUD lawsuits for women who have experienced:
- Fractured or Broken Arms During Removal
- Embedded IUD in Uterus
- Perforation of the Uterine Wall
- Migration to Other Parts of the Body
- Hysterectomy or Invasive Surgery to Remove the Copper IUD
All cases are investigated by our law firm under a contingency fee agreement, which means that there are never any out-of-pocket costs to hire a lawyer and we only receive attorney fees or expenses if a recovery or settlement is obtained for your Paragard IUD complications. To find out if you or a loved one may have a case, Request a free consultation and claim evaluation.
Paragard Cooper IUD Side Effects and Removal Complications
The Paragard IUD was first approved in 1984, and has been marketed since 1988 as a form of long-acting birth control that does not contain or release hormones.
Copper wire coiled around the the small, T-shaped IUD are designed to produce an inflammatory reaction as small amounts of copper released into the uterus, which prevents sperm from reaching the egg. The manufacturer claims that this is more than 99% effective at preventing pregnancy, and Paragard is the only copper IUD sold in the United States.
Paragard has been promoted as safe and effective, indicating that women will face fewer side effects than other methods of birth control. However, it appears that the manufacturer withheld important safety information from doctors and consumers.
Paragard Fracture Case Reports
In 2015, independent researchers published the Open Journal of Clinical & Medical Case Reports, outlining problems at one medical facility where Paragard broke or fractured during removal procedures. The researchers described 7 cases involving problems with Paragard T 380A Copper IUD fractures between 2012 and 2014, including:
- 32 year old woman who had the Paragard IUD embedded during an attempted removal 6 years after the birth control was implanted
- 38 year old woman who had a Paragard IUD arm break during removal after 6 1/2 years
- 48 year old woman who had the ring holding the Paragard IUD strings break during attempted removal after 10 years, resulting hysteroscopic removal of the IUD
- 40 year old woman who experienced problems during Paragard removal in the office 7 years after implant, where an embedded IUD arm broke
- 28 year old woman who required hysteroscopic removal of an embedded copper IUD after it was implanted for 7 years
- 31 year old woman who had a Paragard IUD break during office removal, 7 years after it was implanted
- 33 year old woman who had an embedded Paragard IUD arm break during attempted removal 8 1/2 years after it was implanted
At least six out of seven of the reported cases required hysterocscopic removal of the IUD. Five cases had to be managed in the operating room with polyp forceps and manual vacuum extraction for visualized IUDs, and three were not visible on hysterscopy, resulting in the pieces of the Paragard IUD being left in place.
Researchers indicated that the longer the Paragard IUD remains in place may influence the risk of fracture during removal, as all of the cases described involved IUDs that were in place for 6 to 10 years. The findings at this single institution suggest that the Paragard IUD removal problems are likely underreported.
Questions About Paragard Fracture Risk?
For years, women have relied on false and misleading representations about Paragard IUD safety, including indications that the birth control implant is supposed to be completely reversible and allow them to have children in the future. However, increasing evidence suggests that known side effects of the Paragard IUD were not adequately disclosed to consumers or the medical community.
 Manufacturers knew or should have known about the problems with Paragard IUD breakage, as a result of clinical trial data, post-marketing studies, independent case reports and adverse event data.
Manufacturers knew or should have known about the problems with Paragard IUD breakage, as a result of clinical trial data, post-marketing studies, independent case reports and adverse event data.
Rather than providing accurate warnings, critical safety information was withheld from women about the risk that arms of the Paragard IUD may embed in the uterus or break during removal.
If warnings and information has been provided, many women may have elected different types of birth control or doctors may have changed the way they monitored for signs of potential Paragard IUD side effects.
Allegations Raised in Paragard IUD Class Action Lawsuits
The lawyers at Saiontz & Kirk are evaluating potential class action claims and individual Paragard injury lawsuits for women nationwide. According to allegations being pursued, the manufacturers have:
- Failed to Adequately Warn About the Paragard IUD Removal Risks;
- Failed to Investigate Problems with Paragard Fractures and Broken IUD Arms;
- Failed to Issue a Paragard IUD Recall or Update Warning Label Information
- Designed and Sold a Defective and Unreasonably Dangerous Birth Control Implant
- Placed Profits Before the Interests and Safety of Consumers
No Fees or Expenses Unless You Receive a Paragard IUD Settlement
Saiontz & Kirk is a nationally known law firm that is focused exclusively on representing individuals, we do not represent corporations or insurance companies.
Over the course of nearly 50 years, clients of our law firm have collected more than $1 billion in financial compensation and settlements for cases involving product liability, medical malpractice and other personal injury lawsuits,
All Pargard IUD injury cases are being investigated by our law firm on a contingency fee basis. There are never any out-of-pocket costs to hire our lawyers, and we receive no fees or expenses unless a recovery is obtained.
Protect your family and find out if you or a loved one may be entitled to a Paragard settlement.

