Why is there a Wegovy Lawsuit?
Wegovy lawsuits are being pursued for individuals throughout the United States who were hospitalized due to significant gastrointestinal problems or suffered vision problems after using the weight loss drug.
The link between the weight loss drug Wegovy and gastroparesis side effects was not adequately disclosed by Novo Nordisk, even though the drug maker was aware of wide spread reports involving stomach problems from Ozempic, which was introduced several years earlier for treatment of diabetes and contains the same active ingredient, semaglutide.
In addition to gastrointestinal injuries, new medical research has linked the use of Wegovy and Ozempic to an increased risk of vision problems, specifically, non-arteritic anterior ischemic optic neuropathy (NAION), which is a condition characterized by a sudden decrease in vision due to reduced blood flow to the optic nerve.
Lawsuits claim that Novo Nordisk quickly identified the potential profits from Ozempic’s off-label weight loss prescriptions, and rushed to release a subsequent version called Wegovy, which was approved for individuals seeking an obesity treatment. However, the Wegovy formulation for weight loss purposes contains 2.5 times more of the active ingredient, which may increase the risk of health complications and severe gastrointestinal injuries.
Published case reports and adverse events reported to the U.S. Food and Drug Administration (FDA) make it clear that Novo Nordisk knew or should have known about these potential risks, yet failed to conduct thorough research on the semaglutide drugs side effects, placing it’s desire for profits before consumer safety.
Who Qualifies for a Wegovy Lawsuit?
Financial compensation may be available for individuals who received Wegovy injections, and suffered any of the following health problems:
- Gastroparesis (Stomach paralysis)
- Ileus (Intestinal obstruction)
- Gastric Stasis
- Pancreatic Cancer
- Deep Vein Thrombosis
- Pulmonary Aspiration
- Vision Changes/Blindness
- Other gastrointestinal problems
Our lawyers are also pursuing Ozempic lawsuits and Mounjaro lawsuits, for individuals who took the similar diabetes treatment containing tirzepatide and developed gastrointestinal injuries.
To help determine the Wegovy lawsuit settlement amount that you or a loved one may be eligible to receive, request a no-obligation case evaluation today.
2025 Wegovy Lawsuit Update
Following recent studies and case reports linking Wegovy to gastrointestinal problems and vision problems, individuals are now pursuing Wegovy lawsuits over the manufacturer’s failure to warn about the side effects of the GLP-1 receptor agonist drugs.
- January 1, 2025 Update: Judge Marston has issued a scheduling order in the Wegovy and GLP-1 drug litigation, addressing the manufacturers’ filing of motions to dismiss claims based on arguments that federal preemption shields them from failure-to-warn claims. According to the order, drug makers must file their motions by January 24, 2025, asserting that federal law prevented independent updates to drug warning labels. Plaintiffs are required to respond by March 18, with replies due by April 14, after which oral arguments will be scheduled. Fact discovery on general causation must be completed by July 2, 2025, with expert reports exchanged in July and August. Monthly status conferences will begin January 14, and a decision on preemption may come by the end of the year.
- July 2024 Update: Recent research indicates that individuals taking Ozempic and Wegovy may experience an eightfold rise in the likelihood of vision impairments, specifically from non-arteritic anterior ischemic optic neuropathy (NAION). This condition can trigger abrupt alterations in vision and potentially result in permanent blindness.
- February 2024 Update: In response to a motion to consolidate the growing number of Wegovy and Ozempic stomach paralysis lawsuits filed across various U.S. District Courts, the U.S. JPML issued an order on February 2, 2024 calling for the centralization of all GLP-1 gastroparesis lawsuits in the U.S. District Court for the Eastern District of Pennsylvania before U.S. District Judge Gene E.K. Pratter.
- December 2023 Update: As the number of Ozempic and Wegovy lawsuits over undisclosed gastroparesis side effects continue to be filed, a motion to transfer was filed by a group of plaintiffs requesting that all claims filed throughout the federal court system be consolidated for pretrial proceedings before U.S. District Judge James D. Cain, Jr. in the Western District of Louisiana.
- October 2023 Update: In a study published in the Journal of the American Medical Association (JAMA) on October 5, 2023, researchers identified that Ozempic users face a threefold increased risk of stomach paralysis when compared to those using Contrave (bupropion-naltrexone).
- August 2023 Update: The first of what is expected to be many semaglutide lawsuits was filed over gastroparesis side effects in the U.S. District Court for the District of Louisiana on August 2, 2023.
- July 2023 Update: A CNN investigation warned of an increasing number of individuals suffering from stomach paralysis after using Ozempic and Wegovy. The study further highlighted growing indications that the producers of these weight loss medications were aware of the possible adverse reactions.
What is Wegovy?
Wegovy is a weight loss drug formulated by Novo Nordisk that was approved by the U.S. Food and Drug Administration (FDA) in June 2021, as a chronic weight management treatment for children and adults over 12 years of age that have been diagnosed with either;
- Obesity (defined as a Body Mass Index, or BMI, of 30 or greater), or
- Overweight (BMI of 27 or more) in the presence of at least one weight-related comorbid condition.
Examples of these weight-related medical issues include:
- Type 2 diabetes
- Hypertension (high blood pressure)
- Dyslipidemia (abnormal levels of fats in the blood, such as high cholesterol or triglycerides)
- Heart disease
- Sleep apnea
- Non-alcoholic fatty liver disease (NAFLD)
How Does Wegovy Work?
Wegovy is designed to mimic the naturally occurring hormone GLP-1, which is released by the body in the intestines after eating to regulate blood sugar levels and slow down the passage of food through the digestive tract. Wegovy’s mechanism of action in relation to GLP-1 includes the following;
- GLP-1 Production: Naturally, the human intestines produce the GLP-1 hormone in response to food intake.
- Appetite Regulation: One of the primary roles of GLP-1 is to regulate appetite. When GLP-1 levels rise, they send a signal to the brain that induces feelings of satiety, or fullness.
- Wegovy’s Mimicry: Wegovy is a GLP-1 receptor agonist, which means it mimics the activity of the naturally occurring GLP-1 hormone.
- Stimulation of Insulin: Just like natural GLP-1, when Wegovy is introduced to the body, it stimulates the pancreas to produce insulin. Insulin assists in lowering blood glucose levels by facilitating the uptake of glucose by cells.
- Delay in Gastric Emptying: By mimicking the effects of GLP-1, Wegovy delays gastric emptying, meaning it slows down the rate at which food leaves the stomach and enters the small intestine.
Due to its effect on the gastric emptying process, individuals taking Wegovy may experience gastroparesis side effects, such as nausea, vomiting, and abdominal pain. However, the drug manufacturer failed to adequately disclose these risks to users and the medical community.
Is Wegovy Different than Ozempic?
Both Ozempic and Wegovy injections are glucagon-like peptide-1 (GLP-1) receptor agonists. The key differences between the two formulations lie in their approved use, dosing regimens, and concentrations of semaglutide.

Differences Between Wegovy and Ozempic
Ozempic (semaglutide) received FDA approval in December 2017, to treat type 2 diabetes in adults. Ozempic is a once-weekly injectable GLP-1 receptor agonist designed to regulate blood sugar by enhancing insulin production.
It is administered once weekly. The starting dose is usually 0.25 mg per week, which can be increased to 0.5 mg per week after 4 weeks. Depending on the individual patient’s need and how well the medication is tolerated, the dose may be further increased to a maximum of 1.0 mg per week. However, many doctors prescribe Ozempic off-label for weight loss at much higher doses.
Wegovy (semaglutide) was approved specifically for chronic weight management for those who are either overweight or obese. The drug is intended to be used in combination with a reduced-calorie diet and increased physical activity for individuals to lose weight.
Wegovy is administered once a week but follows a different dosing pattern oriented towards weight management using a much larger dose of semaglutide. Beginning at 0.25 mg every week, the dose escalates in increments every month, moving to 0.5 mg, 1 mg, 1.7 mg, and peaking at 2.4 mg.
In addition to sharing similarities in drug formulation, the drugs have both been associated with an increased risk of gastrointestinal side effects such as nausea, vomiting, diarrhea, and constipation. However, these stomach problems may be a greater risk with Wegovy, due to the higher concentrations of semaglutide.
Wegovy Dosing Increases Side Effect Rates
Wegovy and Ozempic are both medications that contain the active ingredient semaglutide, used for the treatment of type 2 diabetes and weight management. However, Wegovy contains an almost 2.5x higher dose of semaglutide than Ozempic. This increased dosage in Wegovy can lead to a higher incidence of gastroparesis side effects and other safety risks. .
Since the GLP-1 hormone acts directly on the stomach muscles by reducing the rate of gastric contractions, Wegovy’s amplified dose has caused individuals to experience a pronounced slowing of gastric emptying, which has caused severe gastroparesis side effects.
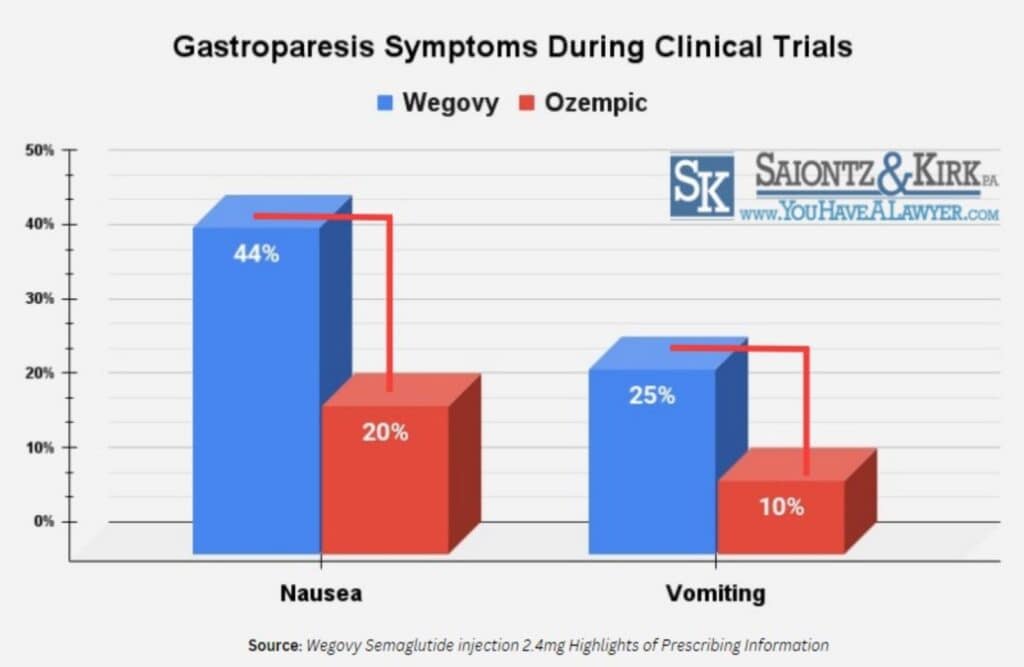
44% of Wegovy Patients Experience Gastroparesis Symptoms
In the clinical trials, approximately 44% of Wegovy users reported nausea, and close to 25% had episodes of vomiting.
On the other hand, 20% of the Ozempic trial participants felt nausea, and 10% indicated instances of vomiting, both of which are common symptoms associated with gastroparesis.
These findings support the theory that the 2.5x greater concentration of semaglutide in Wegovy amplifies the frequency of gastroparesis-related symptoms.
Wegovy and Gastroparesis
Individuals are now filing lawsuits against the makers of Wegovy claiming that the manufacturer was aware of reports of gastric disorders and symptoms that align with the onset of severe gastroparesis, yet failed to further investigate the severity or permanency of the injury risks.
Gastric Disorders From Wegovy
Gastroparesis arises when the stomach muscles malfunction, inhibiting their ability to adequately process and move food. This impedes the stomach from efficiently emptying itself.
Wegovy is intentionally designed to slow gastric emptying to aid in weight loss. As a result, lawsuits claim the manufacturer failed to warn that the continuous use of Wegovy and Ozempic can cause some individuals to experience prolonged and chronic delays in stomach emptying.
Symptoms of Stomach Paralysis from Wegovy
- Nausea
- Vomiting
- Stomach pain
- Abdominal discomfort
- Vomiting undigested food
- Bloating
- Belching
- Heartburn (acid reflux)
- Weight-loss
- Malnutrition
- Stomach obstruction or intestinal blockage
Wegovy Vision Loss Problems
In a study featured in JAMA Ophthalmology on July 3, 2024, researchers highlighted that individuals using Wegovy and Ozempic are up to seven times more likely to experience vision issues, such as blurred or lost vision, compared to those on alternative treatments.
Led by Dr. Jimena Tatiana Hathaway, the research team investigated the connection between Ozempic and a heightened risk of nonarteritic anterior ischemic optic neuropathy (NAION) among people with type 2 diabetes or those who are overweight or obese.
NAION is a disorder marked by a sudden decline in vision resulting from diminished blood flow to the optic nerve. This reduction can lead to lasting damage to the nerve, causing partial or complete vision loss in the impacted eye.
The study assessed data from 16,827 patients reviewed by neuro-ophthalmologists, focusing on individuals with type 2 diabetes or those overweight or obese. It compared the outcomes for those prescribed semaglutide versus those given non-glucagon-like peptide-1 receptor agonist (GLP-1 RA) medications.
Results over a 36-month duration revealed that Ozempic or Wegovy users experienced substantially elevated risks of developing NAION in comparison to those on non-semaglutide medications. Specifically:
- Type II Diabetes Patients: NAION risks rose from 1.8% in non-semaglutide users to 8.9% in semaglutide users, marking a 710% increased absolute risk.
- Overweight or Obese Patients: NAION risks increased from 0.8% in non-semaglutide users to 6.7% among semaglutide users, representing a 590% increased absolute risk.

Studies Linking Wegovy To Gastroparesis
Wegovy lawsuits are pointing to a growing body of research supporting the claims that Novo Nordisk was aware of gastroparesis side effects caused by Wegovy, yet failed to update the prescribing information to warn the medical community or patients.
Wegovy Worsens Stomach Paralysis Symptoms
An August 2020 study named titled Diabetic Gastroparesis: A Review cautioned that drugs like Wegovy, Ozempic, and other GLP-1 diabetes medications could exacerbate stomach paralysis.
“Patients do not know they have diabetic gastroparesis until they are put on a glucagon-like peptide 1 (GLP-1) receptor agonist such as … semaglutide … to manage their blood glucose. … This class of drugs can exacerbate the symptoms of diabetic gastroparesis.”
Wegovy Gastric Emptying Warning
In July 2023, The American Society of Anesthesiologists (ASA) highlighted concerns about delayed gastric emptying caused by Wegovy and Ozempic, especially for those set to undergo surgeries. These side effects, including nausea and vomiting, increase the chance of patients accidentally inhaling food or experiencing choking under anesthesia or deep sedation
Amidst their growing use for weight control, the ASA released guidelines for Wegovy and Ozempic. They advise daily users to avoid these drugs on surgery day, while weekly users should pause a week before surgery. Additionally, if patients experience severe gastrointestinal symptoms, such as acute nausea, vomiting, or abdominal issues, on surgery day, medical professionals should consider rescheduling.
Semaglutide Linked to 3x Risk of Stomach Paralysis
An October 2023, study published in the Journal of the American Medical Association (JAMA) revealed that Ozempic and other semaglutide users face a threefold increased risk of stomach paralysis compared to those on oral weight loss drugs. The research looked at the gastrointestinal side effects reported among users of injectable GLP-1 agonists, semaglutide and liraglutide, and compared the results to those using the oral weight loss drug medication, Contrave.
The findings showed Ozempic had a 1% incidence rate of stomach paralysis, liraglutide had 0.7%, and Contrave had 0.3%. Moreover, the injectable drugs semaglutide and liraglutide presented a fourfold increased risk of bowel obstruction and a ninefold increased risk of pancreatitis compared to Contrave.
Long Term Side Effects of Gastroparesis From Wegovy
Lawsuits claim that when an individual experiences prolonged gastroparesis or stomach symptoms from Wegovy, it can lead to a range of medical complications and the development of other health conditions due to the impaired movement and processing of food in the stomach. Some of the potential health conditions and complications that can arise from long term gastroparesis symptoms include:
- Organ Dysfunction: Consistent vomiting can result in a significant loss of fluids, leading to dehydration. Dehydration can impact kidney function and electrolyte balance, leading to symptoms like dizziness, dry mouth, and rapid heartbeat.
- Deficiencies: Delayed gastric emptying can mean that food is not being properly digested and nutrients aren’t being absorbed effectively. This can lead to deficiencies in essential vitamins and minerals, which can affect almost every system in the body, leading to weakness, fatigue, and decreased immune function.
- Gastroesophageal Reflux Disease (GERD): With gastroparesis, food remains in the stomach for extended periods. This can cause reflux, where the stomach’s acidic contents flow backward into the esophagus. GERD can lead to esophagitis, or inflammation of the esophagus, which may present as heartburn, chest pain, and difficulty swallowing.
- Bezoars: A bezoar is a hard, solid mass of food that forms in the stomach and can cause blockages. These can lead to nausea, vomiting, and can become dangerous if they obstruct the passage of food into the small intestine.
- Blood Sugar Fluctuations: Delayed gastric emptying can cause unpredictable digestion and absorption of food, leading to erratic blood sugar levels. For diabetics, this can make glucose management very challenging, increasing the risk of both hypoglycemia (low blood sugar) and hyperglycemia (high blood sugar).
- Bacterial Overgrowth: When food remains in the stomach for too long, it can allow bacteria to proliferate, which can lead to bacterial overgrowth in the stomach and small intestine. This can result in symptoms like bloating, diarrhea, and abdominal pain.
- Mental Health Impact: Chronic symptoms and the lifestyle adjustments required to manage them can lead to anxiety, depression, and decreased quality of life. The uncertainty of meal-related symptoms can cause stress, and the physical discomfort can lead to mood changes and isolation.
Wegovy Side Effects Reported to the FDA
Novo Nordisk and its subsidiaries put profits before its customers’ safety by ignoring the growing number of side effects that were reported to the FDA by individuals taking Wegovy, lawsuits allege.
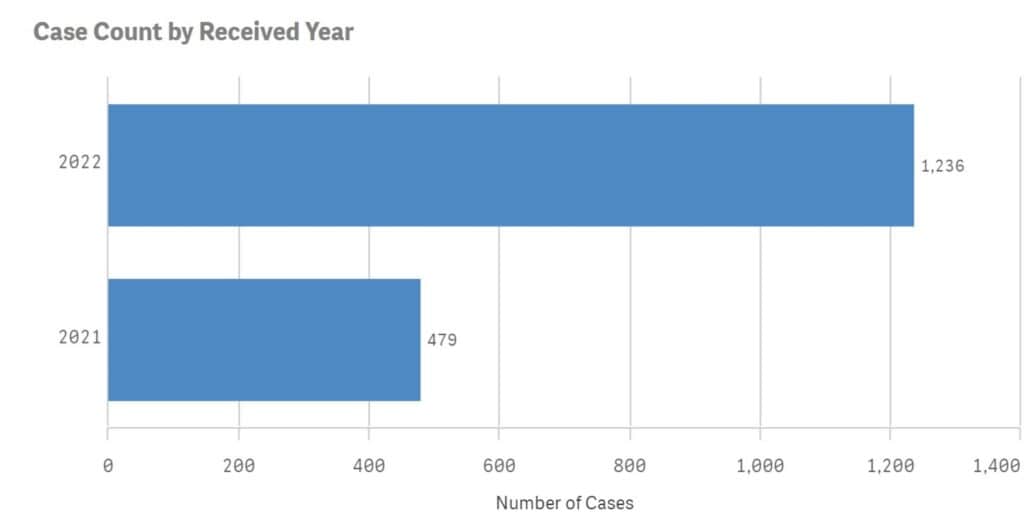
According to the FDA’s FAERS database, the number of adverse reactions each year since Wegovy was released have climbed significantly, with the number of reports nearly tripling by the second year the drug entered the market.
Wegovy Warning Label Failure
Lawsuits claim the manufacturer knew or should have known about the gastroparesis side effects from semaglutide drugs during the initial years, pointing to nearly 20,000 adverse events reports submitted to the FDA for all three versions of semaglutide.
According to the FDA’s database, the number of adverse events reported for each semaglutide drug since they have been released up until June 30, 2023, are as follows;
- Ozempic adverse events: 14,149 (approved 2017)
- Rybelsus adverse events: 2,540 (approved 2019)
- Wegovy adverse events: 2,201 (approved 2021)
As of late 2023, the manufacturer has neither released any patient safety updates nor initiated a Wegovy recall related to the reported gastroparesis side effects. The current labeling continues to downplay the gastrointestinal issues being reported by users.
Rather than taking the necessary action to warn doctors and patients, Novo Nordisk continues to pump billions of dollars into mass manufacturing the drugs and advertising them as safe.
Wegovy Stomach Paralysis Lawsuit Settlement Benefits
A Wegovy settlement may include compensation for various damages suffered due to the side effects of the medication. While specific benefits can vary depending on the case and settlement terms, typically, such benefits can include:
- Medical expenses related to the treatment of side effects experienced from using Wegovy.
- Lost wages if the side effects caused you to miss work.
- Pain and suffering and emotional distress due to the adverse effects of the drug.
- Any other losses or damages that can be directly attributed to the side effects of Wegovy.
Each case is unique, and the actual settlement benefits you may be eligible for would depend on the specifics of your situation. To determine the potential value of your Wegovy lawsuit settlement, contact our lawyers for a free consultation.
Are there any costs to hire a Wegovy Lawyer?
There are absolutely no out-of-pocket costs to review your case or hire our attorneys. Wegovy lawsuits are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis.
Through the use of contingency attorney fees, individuals have can seek compensation through an Ozempic, Wegovy or Rybelsus lawsuit and use the experience and resources of our national law firm — regardless of their individual financial resources.
You pay nothing up front to hire our Wegovy lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturer. Our law firm receives nothing unless we win your case!
What are the steps in an Wegovy case evaluation?
Complete Our Case Evaluation Request Form. Providing contact information and some information about your Wegovy problems.
Get Contacted by Saiontz & Kirk, P.A. You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.

