Spinal Cord Stimulator Lawsuit Status
Spinal cord stimulator lawsuits are being pursued on behalf of individuals who experienced serious complications after receiving a spinal cord stimulation (SCS) device for pain management. For many recipients, the procedure was recommended after years of chronic back pain or nerve pain, making the failure of the implanted device especially devastating.
In many of these cases, the spinal cord stimulator problems center on the lead wires, the thin insulated wires placed near the spinal cord that deliver electrical signals to block pain. Lawsuits allege that some of these lead components may have been improperly manufactured, leading to premature fracture, migration out of position, loss of pain relief or painful electrical shocks. When these failures occur, patients often require additional surgery to reposition or remove the device.
As the number of lawsuits continues to grow, plaintiffs have begun seeking to coordinate the litigation in federal court. On February 20, 2026, a motion was filed with the U.S. Judicial Panel on Multidistrict Litigation requesting that spinal cord stimulator lawsuits involving Abbott Laboratories and Boston Scientific be consolidated before one judge for coordinated pretrial proceedings. The filing indicates that at least 15 related cases are already pending in federal courts nationwide, each raising similar allegations involving device malfunctions, lead failures and neurological injuries. If the JPML grants the request, the cases would proceed as a multidistrict litigation, allowing discovery and other pretrial matters to be managed in a single court as additional claims are filed.
The spinal cord stimulator injury lawyers at Saiontz & Kirk are evaluating product liability claims against major medical device manufacturers including Abbott, Boston Scientific, Medtronic, Nevro, St. Jude Medical and others. Individuals who required revision or removal surgery, experienced worsening chronic pain or suffered electrical shocks after implantation may be eligible to pursue a spinal cord stimulator lawsuit if the device malfunction was linked to a manufacturing defect.
Who Can File a Defective Spinal Cord Stimulator Lawsuit? Individuals may qualify to file a spinal cord stimulator lawsuit if they received an SCS implant and later experienced problems such as:
- Lead wire fracture
- Lead wire migration
- Device migration within the spinal canal
- Electrical shocks or overstimulation
- Worsening chronic pain
- Severe nerve pain
- Spinal cord compression
- Infection
- Device removal or explant procedure
- Permanent nerve damage
- Spinal cord injuries
- Wrongful death
If you or a loved one suffered serious complications after receiving a spinal cord stimulator implant, you may be eligible for a spinal cord stimulator lawsuit settlement. Contact our spinal cord stimulator lawyers today for a free case evaluation. There are no fees unless compensation is recovered.

SCS Lawsuit Info On This Page
What Is a Spinal Cord Stimulator?
Lead Wire Failures and Device Migration Risks
Spinal Cord Stimulator Side Effects and Symptoms
Spinal Cord Stimulator Manufacturers Named in Lawsuits
Abbott Spinal Cord Stimulator Lawsuit
Boston Scientific Spinal Cord Stimulator Lawsuit
Medtronic Spinal Cord Stimulator Lawsuit
Spinal Cord Stimulator Lawsuit Compensation
Evidence Needed to File a Spinal Cord Stimulator Lawsuit
Spinal Cord Stimulator Lawsuit Statute of Limitations
Cost to Hire a Spinal Cord Stimulator Lawyer
Spinal Cord Stimulator Lawsuit FAQs
What Is a Spinal Cord Stimulator?
A spinal cord stimulator is a small medical device that doctors implant in the body to help manage chronic pain. It is usually recommended for people who have tried injections, physical therapy, medications or even back surgery and are still living with daily pain.
The device is placed under the skin, usually in the lower back or upper buttock area. Thin wires called leads are inserted near the spinal cord, and those wires send mild electrical pulses that interrupt pain signals before they reach your brain.
Modern devices often include rechargeable spinal cord stimulators, wireless programming features and adjustable stimulation settings designed to customize pain relief.
How Does a Spinal Cord Stimulator Work?
Your nerves carry pain messages from your back or legs up through your spinal cord to your brain. A spinal cord stimulator works by sending small electrical signals through the lead wires to “mask” or disrupt those pain messages.
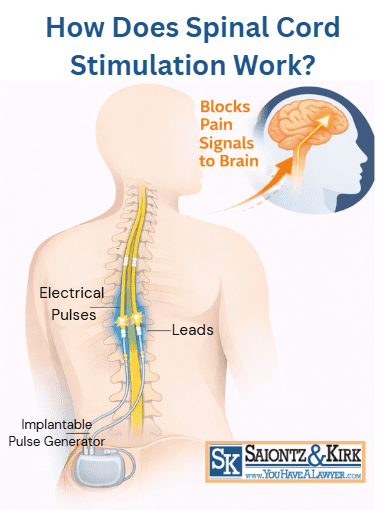
The goal is not to fix the injury itself. Instead, the device is meant to reduce how much pain you feel so you can move better, sleep better and rely less on pain medication.
What Conditions Are Spinal Cord Stimulators Used to Treat?
Spinal cord stimulation devices are often used to treat severe injuries such as:
- Chronic back pain
- Chronic nerve pain
- Failed back surgery syndrome
- Complex regional pain syndrome
- Long-term pain after work injuries
Most people first go through a trial period where temporary wires are placed to see if the device helps reduce their pain. If the trial provides relief, a permanent spinal cord stimulator is implanted during surgery.
However, a growing number of reports have identified SCS lead wire fractures and migrations occurring after permanent implantation, sometimes requiring additional surgery to correct the problem.
Spinal Cord Stimulator Lead Wire Failures and Device Migration
Spinal cord stimulator lawsuits often center on problems involving the device’s lead wires. These thin, insulated wires are placed in the epidural space near the spinal cord and are responsible for delivering the electrical pulses that block pain signals.
Since the entire system depends on these leads functioning properly, even minor problems in how they are manufactured can cause the device to stop working correctly. When a lead fails, the stimulator cannot deliver consistent electrical impulses, and patients may immediately notice worsening pain or abnormal sensations.
Alleged Manufacturing Defects Involving Lead Components
According to allegations raised in complaints, certain spinal cord stimulator devices may not have been manufactured in strict compliance with required federal quality control standards. These cases do not claim that spinal cord stimulation therapy itself is defective. Instead, they focus on whether specific implanted devices failed because critical components, particularly the lead wires, did not meet approved manufacturing specifications.
When those lead components are not properly manufactured, the results can be mechanical failure inside the body.
For example, if an SCS lead wire fractures, it can interrupt the electrical pathway that delivers stimulation. If a lead migrates, meaning it shifts from its original position near the spinal cord, the electrical pulses may no longer target the intended nerves. Patients may then experience sudden loss of pain relief, jolting electrical shocks, burning sensations or worsening chronic pain.
Complaints have identified several types of alleged manufacturing problems involving spinal cord stimulator lead components, including:
- Structural weaknesses that may cause premature lead wire fracture
- Improper anchoring or stabilization that allows leads to migrate out of position
- Insulation defects that permit unintended electrical discharge
- Inconsistent internal conductivity resulting in therapy interruption
- Failures to properly validate or monitor manufacturing processes

Some complaints further allege that certain devices may have deviated from approved manufacturing processes governing firmware execution, electrical stability or component validation. In these situations, the stimulator may shut off unexpectedly, deliver inconsistent stimulation or require repeated reprogramming before ultimately failing.
These mechanical failures often lead to the spinal cord stimulator side effects and neurological symptoms described in the next section.
Spinal Cord Stimulator Side Effects and Symptoms
When a spinal cord stimulator malfunctions, individuals may experience serious side effects that go beyond normal post-surgical discomfort. These complications often occur when lead wires fracture, migrate or fail to deliver consistent electrical impulses to the spinal cord.
In many spinal cord stimulator cases, individuals report worsening symptoms months after implantation, particularly when the implanted device begins to malfunction.
Common Spinal Cord Stimulator Complications and Symptoms
Patients who experienced spinal cord stimulator complications have reported:
- Worsening chronic pain
- Severe nerve pain
- Debilitating back pain
- Sudden loss of pain relief
- Electrical shocks or jolting sensations
- Overstimulation or burning sensations
- Spinal cord compression
- Spinal nerve root irritation
- Muscle weakness
- Numbness or tingling in the limbs
- Infection at the implant site
- Swelling or inflammation near the spinal canal
- Developed chest pain related to device malfunction
- Headaches or neurological symptoms
- Abnormal device behavior during charging
- Therapy shutting off unexpectedly
In more serious cases, individuals have required revision surgery to reposition fractured leads or remove the device entirely. Some underwent multiple surgeries after continued malfunction.
Since spinal cord stimulators are intended to manage chronic intractable pain, the loss of therapy combined with worsening pain can significantly impact daily function, mobility and quality of life can decline quickly.
For some patients, spinal cord stimulator injuries have resulted in permanent nerve damage or long-term spinal cord complications.
Spinal Cord Stimulator Manufacturers Named in Lawsuits
Spinal cord stimulator lawsuits are being pursued against several major medical device manufacturers. Rather than focusing on general design concerns, many of these cases center on allegations that certain spinal cord stimulation devices were not manufactured in accordance with FDA-approved specifications and quality system requirements, particularly with respect to lead wire integrity and device performance consistency.
Abbott Spinal Cord Stimulator Lawsuit
Spinal cord stimulator lawsuits are being filed against Abbott Laboratories alleging that certain Abbott spinal cord stimulator devices, including systems marketed under the Eterna and Proclaim platforms, may have deviated from approved manufacturing standards.
While some of these devices were originally sold as St. Jude spinal cord stimulators, Abbott acquired St. Jude Medical in January 2026 for approximately $25 billion, assuming liability for failures linked to St. Jude SCS devices manufactured before the sale.
According to allegations raised in recent complaints, some Abbott systems and St. Jude systems failed to consistently deliver stable therapy and may have been manufactured in a way that did not comply with federal quality standards.
Plaintiffs claim the devices experienced problems involving internal firmware controls, lead components, and battery or charging systems. These alleged defects have been linked to loss of pain relief, painful electrical shocks, unexpected therapy shutdowns, and lead migration that required additional surgeries.
Abbott Spinal Cord Stimulator Problems
- Lead wire migration requiring revision surgery
- Loss of therapeutic pain relief
- Unintended electrical impulses
- Battery instability or charging failures
- Worsening chronic pain
- Multiple revision surgeries and device removal
Abbott Spinal Cord Stimulators Under Investigation
- Eterna™ SCS System
- Proclaim™ Plus SCS System
- Proclaim™ XR SCS System
- Prodigy MRI™ SCS System (historical/legacy platform)

Boston Scientific Cord Stimulator Lawsuit
Lawsuits have also been filed against Boston Scientific involving spinal cord stimulation systems marketed under names such as the WaveWriter Alpha and related platforms.
According to allegations raised in these Boston Scientific spinal cord stimulator lawsuits, certain systems were substantially modified over time through regulatory supplements without undergoing full independent clinical review, and may have been manufactured in ways that failed to comply with federal quality requirements.
Lawsuits allege the WaveWriter Alpha devices incorporated changes to lead designs, battery architecture, firmware programming, and multiwaveform stimulation systems that altered the safety and performance profile of the original approved device. The complaints further contend that manufacturing defects and inadequate quality controls contributed to lead instability, battery problems, stimulation irregularities, and therapy failure.
Boston Scientific Spinal Cord Stimulator Problems
- Lead wire fracture
- Lead migration requiring revision surgery
- Painful or unpredictable shocking sensations
- Ineffective stimulation despite repeated reprogramming
- Battery instability or battery “flipping” within the pocket
- Device malfunction leading to explant procedures
Lawsuits also allege violations of federal manufacturing and post-market safety regulations, including failures to properly validate design changes, investigate device complaints, implement corrective actions, and update safety labeling.
Boston Scientific Spinal Cord Stimulators Under Investigation
- WaveWriter Alpha™ SCS System
- WaveWriter Alpha™ Prime SCS System
- Spectra WaveWriter™ SCS System
- Precision™ System (legacy)
- Precision Spectra™ System
- Precision Novi™ System
- Precision Montage™ MRI SCS System

Medtronic Spinal Cord Stimulator Lawsuit
Medtronic spinal cord stimulator lawsuits are being filed involving implantable neuromodulation systems marketed under product families such as Restore, RestoreAdvanced, PrimeAdvanced, Intellis, Inceptiv, and earlier Itrel and Synergy platforms.
Complaints allege that certain Medtronic spinal cord stimulation devices experienced manufacturing and quality control deficiencies affecting device reliability and stimulation performance. According to lawsuit investigations, failures in design validation, process controls, and post-market corrective action may have contributed to erratic electrical output, unintended shocks, device migration, and loss of therapeutic benefit.
Medtronic Spinal Cord Stimulator Problems
- Lead fracture
- Device migration
- Electrical overstimulation
- Severe or worsening pain
- Spinal nerve irritation
- Revision surgery or device removal
Lawsuits focus on whether certain Medtronic spinal cord stimulator devices deviated from FDA-approved manufacturing specifications and quality system regulations, resulting in serious complications and additional spinal cord stimulator surgery.
Medtronic Spinal Cord Stimulators Under Investigation
- Intellis™ SCS System
- Intellis™ Pro SCS System
- Inceptiv™ SCS System
- Vanta™ SCS System (recharge-free)
- Restore
- RestoreAdvanced
- RestoreSensor
- Itrel II and Itrel III
- Synergy

What Compensation Is Available in a Spinal Cord Stimulator Lawsuit?
Individuals who file a spinal cord stimulator lawsuit may be entitled to financial compensation for the harm they suffered. Spinal cord stimulator settlements and lawsuit verdicts may include damages for both economic and non-economic losses.
Potential compensation may include:
- Past medical expenses
- Future medical treatment and surgical costs
- Revision surgery or explant procedures
- Lost wages
- Reduced earning capacity
- Pain and suffering
- Permanent nerve damage
- Long-term disability
- Loss of quality of life
- Wrongful death damages for surviving family members
Because spinal cord stimulators are implanted medical devices, complications often require additional surgeries and extended recovery time. Individuals who experienced severe injuries linked to a defective spinal cord stimulator may be eligible to pursue compensation through a defective medical device lawsuit.
What Evidence Is Needed to File a Spinal Cord Stimulator Lawsuit?
If you experienced complications after a spinal cord stimulator operation, certain medical records can help support your case. However, you do not need to gather everything yourself before contacting a lawyer.
An attorney can help obtain the necessary documentation once your case is reviewed. The most important first step is simply reaching out for a free case evaluation.
Spinal Cord Stimulator Records
- Medical records related to your spinal cord stimulator implantation
- The name of the device manufacturer, if known
- Records of revision surgery or device removal
- Imaging showing lead migration or fracture
- Notes describing worsening pain or electrical shocks
Even if you do not have this information readily available, you may still qualify for a spinal cord stimulator lawsuit. Experienced product liability attorneys can work directly with your healthcare providers to obtain operative reports, device details and other necessary documentation.
You do not need to have all the answers before calling. A free case review can help determine whether your spinal cord stimulator complications may be linked to a defective device.
How Long Do I Have to File a Spinal Cord Stimulator Lawsuit?
The deadline to file a spinal cord stimulator lawsuit varies by state and depends on the specific circumstances of the case. In many states, product liability claims must be filed within three years from the date the injury occurred or was discovered.
Because spinal cord stimulator complications may develop months or even years after implantation, determining when the statute of limitations begins to run can be complex. Individuals who experienced worsening chronic pain, lead wire fracture or revision surgery should consider having their case reviewed as soon as possible to protect their legal rights.
Delays in filing a defective medical device lawsuit may prevent recovery, so it is important to act promptly.
How Much Does It Cost to Hire a Spinal Cord Stimulator Lawyer?
There are absolutely no out-of-pocket costs to review your case or hire our attorneys. Potential claims are evaluated for individuals throughout the United States, and all cases are handled on a contingency fee basis.
Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm to pursue a Spinal Cord Stimulator lawsuit settlement — regardless of their individual financial resources.
You pay nothing up front to hire our lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturers. Our law firm receives nothing unless we win your case!
What are the steps in a Spinal Cord Stimulator case evaluation?
Complete Our Case Evaluation Request Form. Providing contact information and some information about your spinal cord stimulator injury case.
Get Contacted by Saiontz & Kirk You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.
FIND OUT IF YOU QUALIFY FOR A SPINAL CORD STIMULATOR SETTLEMENT
Spinal Cord Stimulator Lawsuit FAQs
Is there a spinal cord stimulator lawsuit?
Spinal cord stimulator lawsuits are being pursued by individuals who experienced serious complications after implantation. Many of these claims involve allegations that certain devices were not manufactured in compliance with required federal standards, particularly involving lead wire integrity and device performance.
What is the average spinal cord stimulator lawsuit settlement?
There is no fixed average settlement amount. Compensation depends on the severity of the injury, whether revision surgery was required, the extent of permanent nerve damage, lost income and other long-term effects.
Which spinal cord stimulator manufacturers are facing lawsuits?
Lawsuits have been pursued against manufacturers including Abbott, Boston Scientific, Medtronic, Nevro, St. Jude Medical and others. Claims often focus on alleged manufacturing defects affecting lead wires and implanted components.
Can I file a lawsuit if my spinal cord stimulator was removed?
Yes. Many spinal cord stimulator lawsuits involve individuals who required revision surgery or complete device removal due to lead migration, fracture or device malfunction.
What happens if a spinal cord stimulator lead migrates?
If a lead wire moves out of position, the device may stop delivering proper stimulation. This can cause worsening pain, electrical shocks or loss of therapy and often requires additional surgery to reposition or replace the leads.
Can a spinal cord stimulator make pain worse?
Some patients report worsening chronic pain or new nerve pain after device malfunction. Lead failure or inconsistent electrical output can cause abnormal stimulation patterns instead of pain relief.
How long does a spinal cord stimulator lawsuit take?
The timeline varies depending on the complexity of the case and whether coordinated litigation develops. Some product liability claims resolve within months, while others may take longer depending on court proceedings.
What if I don’t know which device was implanted?
You can still contact an attorney for a free case review. Your medical records typically contain the manufacturer and model information, and an attorney can help obtain that documentation.
Can family members file a wrongful death claim involving a spinal cord stimulator?
In certain circumstances, surviving family members may pursue a wrongful death claim if a defective spinal cord stimulator contributed to fatal complications. Eligibility depends on state law and the specific facts of the case.
How much does it cost to hire a spinal cord stimulator lawyer?
All spinal cord stimulator lawsuits are handled by our law firm on a contingency fee basis. That means that there are no upfront costs, and we receive no legal fees or expense reimbursement unless compensation is recovered.

