Mirena Warnings on IUD Birth Control Movement Risks are Inadequate
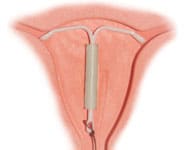
The number of Mirena birth control lawsuits filed throughout the United States continues to mount, with thousands of women alleging that Bayer failed to adequately warn about the potential risks associated with receiving the IUD, specifically the risk of the birth control implant moving long after it is put in place, perforating the uterus and causing serious internal injuries.
Bayer has provided inaccurate and misleading information about the safety of Mirena for both consumers and the medical community. As a result, financial compensation may be available for injuries that may have been avoided if better Mirena warnings had been provided, including information about the risk of:
- Mirena Perforating the Uterus
- Movement of the Mirena IUD to Other Areas of the Body
- Infections
- Complications Requiring Surgical Removal of the Mirena IUD
Mirena IUD Birth Control Warnings
In response to the mounting Mirena litigation, Bayer has attempted to argue that information was provided on the warning label about the risk of perforation. However, the current warnings provided by the drug maker do not accurately disclose the true IUD risks.
When considering whether to use Mirena birth control or another method of contraception to avoid pregnancy, individuals should be provided complete and correct information.
By withhold information about the serious risk that Mirena IUD birth control may spontaneously move months or even years after it is implanted, women were deprived of the opportunity to make an educated decision about whether to receive the Mirena implant.
Mirena birth control warnings suggested that “perforation or penetration of the uterine wall or cervix may occur during insertion.” (emphasis added) However, a substantial number of doctors and women have reported experiencing complications with Mirena IUDs, even after placement was checked during multiple follow up visits after the initial insertion.
Mirena Marketing Claims
Unfortunately, Bayer has a long history of making misleading and false statements about the risks associated with their birth control products.
The decision not to accurately warn about Mirena IUD risks appears to be part of the drug maker’s marketing plan to promote the birth control implant as a hassel free way of avoiding daily birth control pills and making users “feel great.”
In December 2009, Bayer received a warning letter from the FDA over its misleading Mirena claims. The letter focused on a direct-to-consumer Mirena marketing campaign which claimed, among other things, that Mirena IUD birth control had the power to make women feel better about being romantic, help them “reconnect” with themselves and their partners, and help women be more intimate.
In the letter, the FDA stated:
“While we note that Mirena does not involve a daily routine and is not a barrier method of contraception, FDA is not aware of any evidence that suggests that women using Mirena for birth control experience an increase in reconnection, romance, or intimacy with their partners. Claims that state or suggest such quality of life outcomes, such as those described above, must be supported by substantial evidence, as demonstrated through adequate and well-controlled trials using validated patient assessment instruments to measure the outcomes of interest.”
The FDA pointed out that in fact a good number of women reported experiencing decreased libido from Mirena side effects. The agency also pointed out that some women suffered abdominal pain, nausea, headache, nervousness and a depressed mood, “which could adversely affect a woman’s feelings relating to romance or intimacy.”
The FDA also criticized the company for claiming Mirena would help women look and feel great:
“Patients using Mirena may experience various side effects, such as irregular bleeding, ovarian cysts, back pain, weight increase, breast pain/tenderness, and acne, in addition to the side effects indicated above. The experience of these side effects can prevent patients from ‘looking and feeling great.'”
Certainly accurate warnings about the risk of the IUD perforating the uterus years and moving to other parts of the body would have taken away from the marketing message Bayer was trying to build.
Bayer faced similar criticism over marketing of their Yaz and Yasmin birth control pills. The FDA forced the drug maker to spend an unprecedented $20 million on a corrective advertising campaign to correct false and misleading statements in earlier ads. The drug maker has now paid about $2 billion to settle Yaz and Yasmin lawsuits brought by women who allege severe blood clot injuries could have been avoided if accurate warnings had been provided. All of these expenses could have been avoided if Bayer had adequately researched their birth control product and fully disclosed the potential risks in warnings provided to women and doctors.
Mirena IUD Birth Control Movement Lawsuits
The Mirena injury lawyers at Saiontz & Kirk represent women throughout the United States who required surgical removal of the IUD birth control after it moved months or years after being implanted. All cases are handled under a contingency fee agreement, which means that there are no attorney fees unless a recovery is obtained.
To obtain a free and confidential case evaluation, request a free consultation or call 800-522-0102.


2 Comments • Add Your Comments
Lena says:
I had the Mirena. 3 yrs in i was told it must of fell out, dr. Sent me for ultra sound and found it ( not where it was suppose to be with no string attached) I had to have it surgically removed whiched caused me to have a partial hysterectomy.
Posted on November 19, 2019 at 7:32 pm
Lena says:
I had the Mirena. 3 yrs in i was told it must of fell out, dr. Sent me for ultra sound and found it ( not where it was suppose to be with no string attached) I had to have it surgically removed whiched caused me to have a partial hysterectomy.
Posted on November 20, 2019 at 12:58 pm