Breast Mesh Side Effect Lawsuit Overview
Breast mesh lawsuits are being investigated on behalf of women who underwent internal bra procedures and later developed serious side effects or complications that required revision or removal surgery.
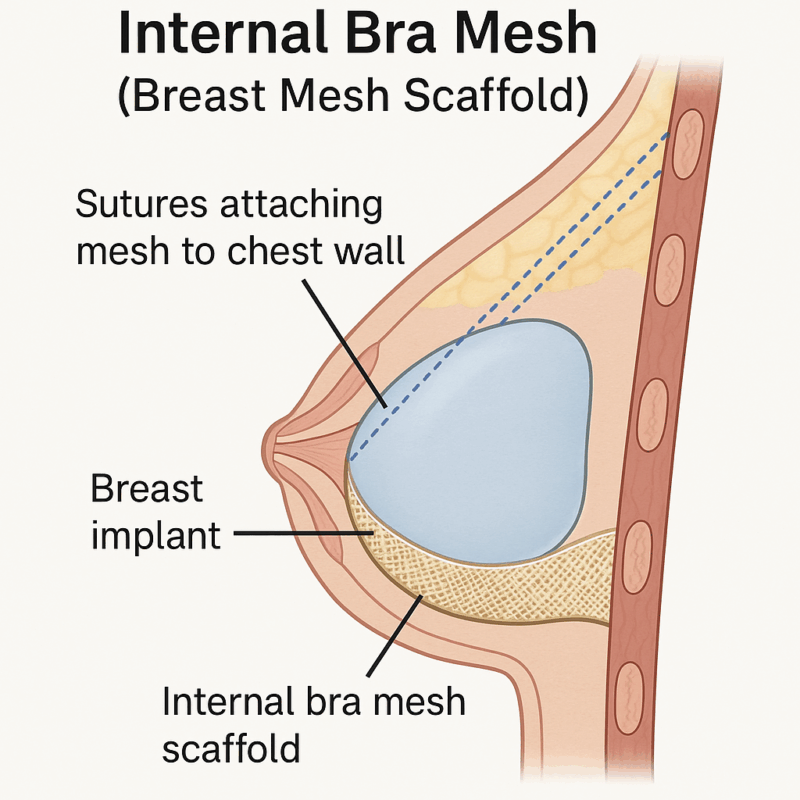
Often referred to as breast mesh scaffolds, these surgical implants are made from materials originally designed for hernia repair and other soft-tissue reinforcement. In recent years, they have been repurposed and sold to plastic surgeons as internal support slings intended to maintain breast shape and stability following augmentation, lift, or reconstruction procedures.
Although manufacturers sold these products that were used off-label for breast mesh implants and post-mastectomy breast surgeries, none have been approved or cleared by the U.S. Food and Drug Administration (FDA) for safe or effective use in breast tissue.
With a growing number of women reporting painful complications, GalaFLEX lawsuits and Phasix lawsuits are now being pursued over manufacturers alleged failure to adequately warn about the risks of using these materials in the breast.
Lawsuits allege manufacturers placed a desire for profits before the health and safety of women, who are often left with disfiguring injuries and the need for multiple corrective surgeries.
Who Qualifies for a Breast Mesh Lawsuit?
Financial compensation through a breast mesh lawsuit settlement may be available for women who received a surgical mesh and later developed any of the following complications:
- Infection or abscess formation
- Chronic pain or nerve injury
- Mesh migration, detachment, or displacement
- Seroma or persistent fluid buildup
- Implant rupture or reconstruction failure
- Skin erosion or tissue necrosis
- Scarring or deformity requiring surgery
There are no fees or expenses unless a settlement or recovery is obtained.
Index of Breast Mesh Side Effect Info On This Page
How Breast Mesh Is Used for Augmentations
Why Breast Mesh Can Fail Inside the Breast
Breast Mesh Side Effects and Complications
Studies Linking Breast Mesh to Serious Injuries
Common Breast Mesh Side Effect Questions
Are there any costs to hire a Breast Mesh lawyer?
How Internal Bra Mesh Is Used in Cosmetic and Reconstructive Surgery
Many women are only learning after the internal bra technique was used during their breast surgery that it may be the source of their ongoing pain or complications.
Synthetic internal bra mesh, such as the GalaFLEX and Phasix mesh are typically inserted beneath or around the breast implant to act as a sling, holding the breast in place and reducing pressure on skin and sutures. The technique was used by surgeons as a way to enhance lift, projection, and the longevity of cosmetic procedures such as breast augmentation, lift, reduction, or revision surgeries.
However, lawsuits now highlight that the breast is a far more dynamic environment than the abdomen or groin, where many mesh products were originally used. Inside the breast, mesh can stretch, degrade, migrate, or fail to integrate properly.
As a result, many women have reported severe side effects such as chronic swelling, tissue hardening, infection, and implant failure, often requiring additional surgeries to remove or replace the mesh.
Breast Mesh in Elective Cosmetic Surgery
In elective procedures, internal bra mesh is often used to add structure and support beneath the skin, especially in women with thin tissue or a history of sagging. Surgeons may insert the mesh to reinforce the implant pocket, stabilize results, and prevent complications like “bottoming out,” where the implant drops too low.
These implants have been commonly used during:
- Breast augmentation, to hold the implant in place
- Breast lift (mastopexy), to support the lifted tissue
- Breast reduction, to reshape and stabilize the breast
- Revision surgeries, after prior implants or cosmetic work failed
Although the mesh was intended to integrate with the body, the breast is constantly changing due to hormones, weight, and movement. Over time, many women experienced mesh failure—leading to swelling, hardening, chronic pain, or the need for additional surgeries to remove or replace the material. Lawsuits are now being pursued, alleging that women were not properly warned about these risks from breast mesh.
Mesh Used in Reconstruction After Cancer Surgery
Internal bra mesh has also been widely used in breast reconstruction for cancer survivors, often placed beneath the skin or chest muscle to help shape the new breast and hold an implant or expander in place.
These materials were marketed as natural, body-friendly scaffolds. But when they failed to integrate, the results can lead to:
- Implant loss
- Skin necrosis
- Chronic infections
- Reconstruction collapse
For breast cancer survivors seeking healing and closure, mesh complications have added a painful and unexpected setback. Lawsuits allege that manufacturers knew these materials were not tested or approved for breast reconstruction but pushed them into the market anyway, without proper warnings about the risks.
Why Breast Mesh Fails Inside the Breast
The breast is a complex, constantly moving environment, and mesh materials were never specifically designed for these conditions. Now, women are pursuing lawsuits over the way manufacturers sold these products to plastic surgeons, who used them in breast tissue, despite never receiving FDA approval for that purpose.
Below are several of the reasons outlined in either medical studies or lawsuit investigations that explain why internal mesh products often fail when implanted in the breast.
Delicate and Dynamic Environment
- Constant motion: Daily movement, exercise, and gravity put strain on the mesh and its attachment points.
- Soft, fatty tissue: Unlike firm abdominal muscle, breast tissue offers little structure for the mesh to anchor securely.
Chronic Inflammatory Response
- Foreign material reaction: The body attempts to isolate the mesh, leading to thick scar tissue formation.
- Failed integration: Instead of blending with tissue, mesh can trigger prolonged inflammation that weakens the surrounding area.
- Collagen remodeling issues: The healing process may distort normal tissue architecture, setting the stage for later complications.
Degradation and Migration
- Unpredictable breakdown: Supposedly “absorbable” meshes can degrade unevenly, leaving sharp or stiff fragments behind.
- Loss of stability: Over time, mesh tension changes, allowing it to stretch, curl, or detach from the chest wall.
- Structural collapse: Once the mesh fails, the implant pocket can lose shape or support entirely.
High Risk of Contamination
- Limited blood flow: The internal breast pocket may heal slowly and provide poor immune defense.
- Bacterial adhesion: Even small surface bacteria can form biofilms that resist antibiotics, setting the stage for infection.
Breast Mesh Complications and Side Effects
Adverse event reports submitted to the FDA through its MAUDE database describe a pattern of serious side effects and complications following internal bra mesh implantation, echoing concerns raised in published medical literature and patient accounts.
Women across the country report injuries that often emerge as the mesh degrades, migrates, or contracts inside the breast, leading to chronic pain, cosmetic deformities, infections, and, in many cases, additional surgeries to remove or revise the implant.
Nerve Damage and Chronic Breast Pain
Some women have reported nerve damage or chronic pain after receiving breast mesh during reconstruction, augmentation or revision surgery. These symptoms may develop when mesh, scar tissue or inflammation irritates nerves in the breast, chest wall or underarm area. Nerve-related pain may also worsen after revision surgery if damaged tissue must be removed.
Symptoms: Women may experience burning pain, sharp shooting pain, tingling, numbness, hypersensitivity or pain that radiates toward the arm, shoulder or chest wall. Some report that the pain interferes with sleep, exercise, clothing tolerance or normal daily activity.
Treatment: Treatment may involve pain medication, physical therapy, nerve blocks or other conservative care. However, persistent nerve-related symptoms may require surgical evaluation, particularly if doctors believe the mesh or surrounding scar tissue is contributing to the pain. Even after removal, nerve recovery may take months and may not be complete.
Infection or Abscess
Breast mesh lawsuits are focusing on serious infections or abscesses that develop more than 90 days after the mesh was implanted. Delayed infections may raise concerns that bacteria attached to the mesh surface, formed resistant biofilms or became difficult to eliminate without surgical removal.
Symptoms: Signs may include redness, warmth, swelling, worsening breast pain, drainage, fever or a firm and tender area around the implant. Abscess formation may cause localized swelling, pus, pressure or worsening pain.
Treatment: Delayed mesh-related infections often require more than antibiotics. Doctors may need to drain the abscess, remove the mesh, remove or replace the breast implant, or perform additional reconstructive surgery after the infection clears.
Seroma and Chronic Fluid Buildup
Seroma occurs when fluid collects around the breast implant, mesh or surgical pocket. In breast mesh cases, women have reported persistent or recurring fluid buildup that required repeated drainage procedures or additional surgery.
Symptoms: A seroma may cause swelling, tightness, tenderness, breast heaviness or a sensation of fluid movement. The breast may appear larger, distorted or uncomfortable. If the fluid becomes infected, redness, fever, drainage or worsening pain may develop.
Treatment: Doctors may drain the fluid with a needle or catheter. If seromas recur or become infected, surgical treatment may be needed to remove the mesh, clean the surgical pocket or revise the reconstruction.
Hematoma
A hematoma is a collection of blood that forms inside the breast pocket after surgery. In breast mesh cases, hematomas may cause swelling, pressure, pain and visible changes in breast shape. Some reports describe hematomas requiring drainage or surgical evacuation.
Symptoms: Women may notice sudden swelling, bruising, firmness, pressure or pain in the affected breast. The breast may become visibly enlarged, tight or discolored.
Treatment: Smaller hematomas may be monitored, but larger or painful hematomas may require drainage or surgery to remove the blood collection. If the hematoma affects the implant pocket or mesh placement, revision surgery may be necessary.
Mesh Migration or Displacement
Mesh migration or displacement may occur when the internal support material loosens, detaches or shifts from its intended position. This can affect the implant pocket and cause visible changes in breast position, shape or symmetry.
Symptoms: Women may notice one breast sitting lower than the other, implant shifting, rippling, bulging, “bottoming out” or visible asymmetry. Some may feel that the breast implant is unstable or no longer properly supported.
Treatment: Corrective surgery is often needed to reposition the implant, remove loose or displaced mesh, repair the implant pocket or rebuild support using other surgical techniques.
Skin Necrosis
Skin necrosis occurs when breast tissue or skin loses blood supply and begins to die. In breast mesh cases, necrosis may be linked to infection, pressure, inflammation, surgical complications or tissue breakdown around the implant and mesh.
Symptoms: The skin may appear dark, discolored, thin, shiny, painful or slow to heal. Open wounds, drainage, exposed mesh or visible implant material may signal serious tissue damage requiring urgent medical care.
Treatment: Treatment may involve wound care, antibiotics, debridement of dead tissue, mesh removal, implant removal or additional reconstructive surgery. Severe cases may result in permanent scarring, deformity or implant loss.
Reconstructive Failure
Reconstructive failure may occur when breast mesh does not provide lasting support, the implant pocket breaks down, the mesh fails to integrate with surrounding tissue or complications make the reconstruction unsustainable.
Symptoms: Women may experience implant dropping, breast asymmetry, loss of support, visible deformity, pain, skin breakdown or repeated complications that prevent the reconstruction from healing properly.
Treatment: Reconstructive failure often requires revision surgery. Doctors may need to remove the mesh, remove or replace the implant, rebuild the breast pocket or perform a new reconstruction. In some cases, women may decide not to undergo further reconstruction after repeated failed procedures.
Implant Rupture
Some breast mesh adverse event reports involve implant rupture or device failure after mesh-supported breast surgery. Implant rupture can cause sudden or gradual changes in breast shape and may be accompanied by pain, swelling or visible asymmetry.
Symptoms: Signs may include breast deflation, shape change, sagging, asymmetry, firmness, pain or the breast falling to the side. Silicone implant ruptures may be less obvious and may require imaging to confirm.
Treatment: Implant rupture typically requires surgical evaluation. Treatment may involve removal of the ruptured implant, removal of damaged mesh, cleaning of the implant pocket and placement of a new implant or alternative reconstruction.
Explant or Revision Surgery
Many women pursuing breast mesh lawsuits report that complications required explant surgery, revision surgery or additional corrective procedures. These surgeries may be needed when mesh causes pain, infection, fluid buildup, tissue damage, implant rupture, loss of support or reconstructive failure.
Symptoms leading to revision: Persistent pain, infection, seroma, hematoma, implant shifting, breast deformity, skin necrosis, rupture or reconstruction failure may all lead doctors to recommend additional surgery.
Treatment: Revision surgery may involve removing the mesh, removing or replacing the implant, draining fluid or blood, repairing tissue damage, rebuilding the breast pocket or correcting deformity. Some women require multiple procedures before the condition stabilizes.
Breast Mesh Removal and Repair Surgery
Women pursuing claims say that once internal bra mesh fails, recovery rarely ends with rest or medication. Most are forced to undergo one or more revision surgeries, sometimes urgently, to remove the mesh, repair damage, and try to restore the breast’s appearance. These breast mesh explant surgeries can be especially overwhelming for women already recovering from cancer treatment or previous cosmetic breast procedures.
Common Surgical Repair Procedures
- Mesh and implant removal: Complete removal of the mesh and breast implant to address infection, inflammation, or other complications.
- Capsulectomy: Surgical removal of thickened scar tissue that has formed around the implant.
- Tissue reconstruction or flap surgery: Rebuilding the breast using the patient’s own skin or muscle, often taken from the back or abdomen.
- Implant replacement or secondary reconstruction: Placement of a new implant after healing, typically delayed to allow tissue recovery.
Studies Linking Breast Mesh to Serious Injuries
Although internal bra mesh implants were sold as a modern advancement for soft tissue repair, multiple independent studies have found that complications may occur when these materials are placed in soft breast tissue, rather than in areas like the abdominal wall where they were originally designed to be used.
Researchers have consistently observed that both synthetic and biologic meshes can contribute to infection, inflammation, and tissue breakdown, leading to implant loss and the need for revision surgeries. These findings help explain why many women are now filing lawsuits after suffering severe complications following breast augmentation or reconstruction procedures involving mesh.
2024 Review Finds Significant Mesh-Related Complication Rates
A 2024 review published in the Aesthetic Surgery Journal Open Forum analyzed 31 clinical studies evaluating synthetic mesh used in implant-based breast reconstruction.
The analysis found that breast mesh complications are not rare — and often serious enough to require surgical correction:
- Reoperation: ~10% of patients required at least one additional surgery to address mesh-related problems
- Infection: ~4% developed postoperative infections involving the mesh pocket or implant
- Seroma (Fluid Buildup): ~3% experienced chronic or recurrent fluid accumulation
- Implant Loss: ~3% suffered complete loss of the implant due to infection or mesh failure
2023 Meta-Analysis Confirms Risks Across Biologic and Synthetic Meshes
A 2023 meta-analysis published in the Aesthetic Surgery Journal examined 24 studies covering more than 2,000 breast reconstructions involving biosynthetic and bioresorbable mesh products. Although many of these devices were marketed as safer, “absorbable” alternatives, complication rates remained concerning:
- Infection ~5%
- Seroma ~5%
- Skin Necrosis ~5.5%
- Implant Loss ~4%
Authors of the study concluded that even bioresorbable mesh products carry risks of infection, inflammation, and reconstructive failure.
What These Studies Reveal About Breast Mesh Safety
Together, these findings demonstrate a consistent pattern across both synthetic and biologic materials:
- The same complication types recur across all mesh products, regardless of brand or composition.
- Complication rates mirror those seen in earlier hernia mesh complications and transvaginal mesh failures, suggesting common design and biological integration issues.
- Revision surgery and implant loss remain among the most common outcomes when mesh fails to integrate properly.
Common Breast Mesh Side Effect Questions
Why does my breast feel hard or painful months after surgery?
- Capsular contracture or mesh inflammation may cause the tissue to tighten around your implant, leading to hardness, pain, or distortion. These symptoms should be evaluated promptly by your surgeon or another qualified plastic surgeon.
Can internal bra mesh cause infections long after surgery?
- Yes. Bacteria can attach to the mesh and form a biofilm that resists antibiotics, leading to delayed or recurring infections even months or years later.
What are the signs that my breast mesh is failing or detaching?
- Common signs include breast asymmetry, sagging, sharp pain, or a feeling that your implant has shifted. Some women also notice visible rippling, fluid buildup, or a sudden change in breast shape.
Can a failed internal bra mesh be removed safely?
- Most cases can be corrected with mesh removal or revision surgery, but the procedure may be complex depending on how much scar tissue has formed. An experienced reconstructive surgeon can evaluate your options.
Is it normal to feel numbness or nerve pain after breast surgery?
- Some nerve irritation is common early in recovery, but persistent tingling, burning, or shooting pain could indicate nerve injury or mesh-related inflammation.
Can breast mesh cause autoimmune or body-wide symptoms?
- In some cases, mesh breakdown particles can trigger immune reactions that cause fatigue, joint pain, or inflammation throughout the body. Many women report improvement after mesh removal.
Will insurance cover mesh removal or corrective surgery?
- Coverage varies. If your procedure was reconstructive after cancer, insurance may help. Cosmetic cases are more challenging, but coverage may apply if infection or medical complications occur.
How long does it take to recover after breast mesh removal?
- Initial healing takes several weeks, but full recovery can take several months depending on tissue damage, infection, and whether additional reconstruction is needed.
Can I still get implants again after mesh complications?
- Yes, but only after your tissue has fully healed. Many surgeons recommend waiting 6–12 months and using natural tissue reconstruction if scar damage is extensive.
Why are women filing breast mesh lawsuits?
- Lawsuits allege that mesh manufacturers sold these devices for breast use without FDA approval, failed to warn about known risks, and caused women unnecessary pain, deformity, and repeat surgeries.
Are there any costs to hire a Breast Mesh Lawyer?
There are absolutely no out-of-pocket costs to review your case or hire our attorneys. Breast mesh claims are evaluated by our lawyers for individuals throughout the United States, and all cases are handled on a contingency fee basis.
Through the use of contingency attorney fees, individuals have access to the experience and resources of our national law firm to pursue a breast mesh lawsuit settlement — regardless of their individual financial resources.
You pay nothing up front to hire our Breast Mesh lawyers, and we only receive an attorney fee or expenses out of the money that is obtained from the manufacturers. Our law firm receives nothing unless we win your case!
What are the steps in a Breast Mesh case evaluation?
Complete Our Case Evaluation Request Form. Providing contact information and some information about your Breast Mesh case.
Get Contacted by Saiontz & Kirk You will be contacted by our law firm to help determine if financial compensation may be available for you and your family.
You Decide If You Want to Move Forward. If our lawyers determine that we can help with your case then you decide whether to move forward and hire us to pursue compensation.
FIND OUT IF YOU QUALIFY FOR A BREAST MESH LAWSUIT SETTLEMENT
