Philips Respironics Recall: Cancer and Health Risks from DreamStation CPAP Foam
April 2024 Update: Following a recent $1.1 billion Philips CPAP settlement to resolve cancer and injury lawsuits, the lawyers at Saiontz & Kirk. P.A. are no longer reviewing new claims.
February 2024 Update: Just two days after Philips officially announced it was halting sales of all its CPAP devices in the United States until 2025 as part of an agreement with the federal government, the U.S. Food and Drug Administration (FDA) released an update on Philips CPAP medical device reports (MDRS), indicating recalled Philips CPAP machines and other breathing assistance devices have been linked to more than 116,000 MDRs and 561 deaths.
September 2023 Update: A $479 million Philips CPAP class action settlement was proposed on September 7, 2023, after the parties submitted a motion to the U.S. District Judge seeking approval of terms to resolve economic loss claims associated with recalled Philips CPAP, BiPAP, and ventilators.
When was the Philips Dreamstation CPAP recall first issued?
On June 14, 2021, Philips Respironics recalled several million DreamStation CPAP machines, BiPAP machines and ventilators sold since 2009. The Philips CPAP recall results from problems with sound abatement foam inside the machines, which may release black debris or toxic chemicals into the airway.
Although the first warnings about foam problems with CPAP machines were disclosed in an investor report in April 2021, the Philips Respironics recall notice was not publicly posted by the manufacturer until weeks later.
An estimated 3.5 million CPAP, BiPAP and ventilator devices were impacted by the recall, each containing a PE-PUR sound abatement foam that is designed to reduce noise and vibrations of the machine. However, the polyester-based polyurethane foam can degrade and release particulate matter and toxic gas into the users’ respiratory airway.
Most of the devices impacted by the Philips Respironics recall are the company’s DreamStation CPAP machines. However, 18 different products sold between 2009 and April 2021 have been recalled.
Which Philips Respironics Machines are being recalled?
A complete list of the recalled Philips Respironics CPAP, BiPap and ventilator machines can be found in an FDA Safety Communication issued on June 30, 2021. Philips Respironics has also released images of the different machines that may contain the defective foam. Check the images below to see if you were using a recalled CPAP machine:
Recalled Philips Respironics CPAP and BiLevel PAP Device Images



Recalled Philips Respironics Mechanical Ventilator Device Images


Images captured from https://www.usa.philips.com/healthcare/e/sleep/communications/src-update
2022 Philips CPAP Recall Updates
Following the 2021 recall involving toxic foam particles in millions of Philips CPAP and ventilator machines, two additional Philips CPAP and BiPAP recalls were issued in 2022.
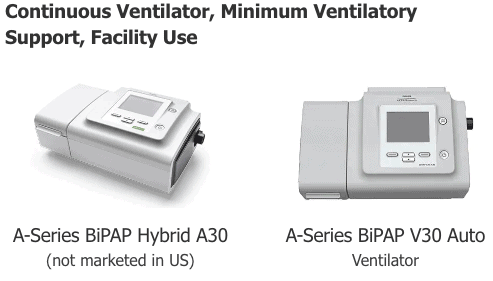
On August 29, the FDA issued a Philips BiPAP recall impacting several models of ventilators that are equipped with a non-compatible material, which may release volatile organic compounds (VOCs) directly into the machine’s air pathways.The recall includes the following models that were manufactured and distributed for sale throughout the U.S. between August 6, 2020, and September 1, 2021;
- A-Series BiPAP A30 (Ventilator)
- A-Series BiPAP A40 (Ventilator)
- A-Series BiPAP V30 (Auto Ventilator)
- OmniLab Advanced+
Officials warned the plastic in the motor could release toxic VOC’s that have could result in a series of health issues including;
- Headache
- Dizziness
- Irritation in the eyes, nose, respiratory tract (airway), and skin
- Hypersensitivity
- Allergic reaction or another immune system reaction
- Nausea or vomiting
- Toxic cancer-causing effects
Just a week later, Philips announced a BiPAP and CPAP mask recall on September 6, 2022, impacting more than 17 million BiPAP and CPAP masks containing magnets that officials warned could interfere with life-saving medical device functionality, such as pacemakers, cardioverter defibrillators and other implantable metallic medical devices.
The Philips BiPAP and CPAP masks impacted in the recall include the devices below;
- Amara View Full Face Mask
- DreamWisp Nasal Mask
- DreamWear Full Face Mask
- Wisp and Wisp Youth Nasal Mask (40lbs or more)
- Therapy Mask 3100 NC/SP (40lbs or more)
As of August 30, 2022, the FDA has received at least 14 Medical Device Reports (MDRs) involving BiPAP and CPAP machine magnetic headgear clips causing serious and potentially life threatening injuries to users, or those with implanted devices nearby. The reports included instances of pacemaker failure leading to pacemaker replacement, a need for shunt adjustment, resetting of automatic implantable cardioverter defibrillators, arrhythmia, cognitive changes, headaches, change in heart rate (tachycardia, bradycardia), convulsions (seizures), and irregular blood pressure.
How do I know if my Philips CPAP has been recalled?
There is no full list of serial numbers impacted by the Philips Respironics recall that has been released. Owners must begin the process of registering their unit with the manufacturer to check whether a Philips CPAP machine has been recalled. That process can be started by clicking here.
Before confirming that the serial number for your sleep apnea machine is included in the recall, it is necessary that you indicate on the first page that you are a “Patient/Device User/Caregiver” and select your country:
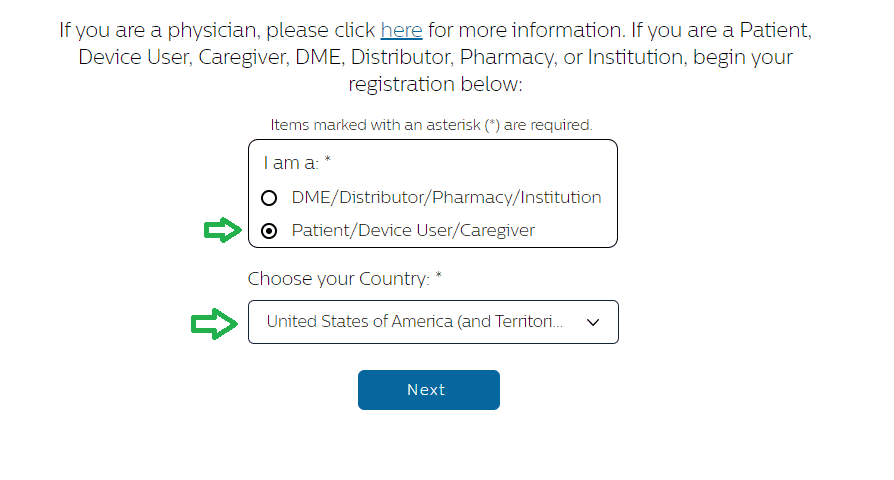
After hitting “Next”, you will be asked to enter your Philips CPAP serial number on the second page to check whether your device is recalled.
The serial number for recalled Philips Respironics sleep apnea machines is printed on a label on the bottom of the device, including a series of letters and numbers that follow the “SN” or “S/N” on the label.
It is important to enter the entire serial number without any spaces, and to make sure you use the number printed on the label of the actual machine, and not any attached water reservoir/tank.
What do I do if I am using a recalled Philips Respironics machine?
Immediately after determining if you or a loved one are using a recalled Philips Respironics machine, you should:
- Stop use of BiLevel PAP or CPAP sleep apnea devices, and contact your physician to discuss treatment alternatives;
- Continue to use life-sustaining ventilators, but consult with your physician as soon as possible about the health risks;
In September 2021, a Philips Respironics recall repair and replacement program was announced by the manufacturer. However, it indicates that the process will likely take over a year, leaving owners without the ability to use their CPAP machine.
Philips CPAP Device Refund and Reimbursement Payments
As part of a settlement agreement reached by Philips and the parties of a class action lawsuit in September 2023, the manufacturer is now offering to pay individuals a set amount of money for the return of their recalled CPAP, BiPAP and ventilator devices.
Philips has agreed to pay class members $100 for each recalled device they return, and an additional fixed amount depending on the device. Below is a list of the amounts individuals may be eligible to receive for each specific recalled model.
- System One 50 Series ASV4 (Auto SV4): $469.14
- System One 50 Series Base: $69.14
- System One 50 Series BiPAP: $159.46
- System One 60 Series ASV4 (Auto SV4): $424.32
- System One 60 Series Base: $68.24
- System One 60 Series BiPAP: $152.70
- C-series S/T, AVAPS (C-series and C-series HT): $394.37
- DreamStation CPAP: $55.63
- DreamStation ASV: $379.50
- DreamStation ST, AVAPS: $329.05
- DreamStation BiPAP: $130.63
- DreamStation Go: $107.43
- E30: $453.83
- OmniLab Advanced Plus: $165.99
- Trilogy 100/200, Garbin Plus, Aeris LiveVent: $1,552.25
- V30 auto: $67.12
Philips has also agreed to allocate up to $15 million to reimburse class members who purchased, rented or leased replacement devices between June 14, 2021, and September 7, 2023. To be eligible for the device reimbursement , individuals must meet the following criteria;
- The new device must have been purchased personally by the claimant.
- The purchase date should fall between June 14, 2021, and September 7, 2023.
- At the time of this purchase, Philips should not have provided the claimant with a repaired or rectified version of their recalled device.
Was there a CPAP Side Effects Lawsuit against Philips?
Yes. The product liability lawyers at Saiontz & Kirk helped individuals who suffered any of the following side effects after using a recalled Philips CPAP, BiPAP or ventilator seek compensation for their injuries;
- Cancer, Lymphoma, Leukemia or Tumors
- Pulmonary Fibrosis
- Interstitial Lung Disease (ILD)
- Pulmonary Fibrosis
- Sarcoidosis
- Pneumoconiosis
- Auto-Immune Disorders
- Chronic Asthma
- Chronic Bronchitis
- Recurrent Pneumonia
- Hypersensitivity
- Kidney Disease
- Liver Disease
- Heart Attack
- Heart Failure
- Stroke
- Wrongful Death


16 Comments • Add Your Comments
Linda says:
Since using my DreamStation I have been coughing up black debris resembling soot. Also have developed bronchitis with a recurring cough.
Posted on July 14, 2021 at 1:42 am
Lawrence says:
Developed a shortness of breath about a year after start of use. Been using for about 3 years. I have a DreamStation machine. I have not been contacted about recall yet.
Posted on July 17, 2021 at 12:34 pm
Leon says:
I would like to speak to someone to review my case
Posted on July 20, 2021 at 11:13 am
Shirley says:
Been on machine for several years. Have developed breathing problems and have severe mucus drainage all day off and on , and continuous daily drainage from my ears. Also coughing up really thick mucus all day where it drains down my throat at night while I sleep.
Posted on July 22, 2021 at 10:37 am
Deborah says:
I just learned of the recall today, by accident, when I heard a recording at my local health care supplier’s business when trying to order a broken part for my machine. Had I not inquired about the reference to a recall on some equipment, I’d never have known about it. Apparently they health care company was supposed to send a certified letter to affected customers. I have used my machine for about 10 months and frequently have coughing when I begin using it at night. It may be the forced air irritating my lungs, but this recall makes me wonder if my lungs are being irritated by off-gassing from a defective machine. So distressing! I do have a call into my dr, as recommended by Philips, but haven’t heard back (just called today) if I should continue to use this machine until they provide a fix of some kind, or if I should stop using it immediately. I’d appreciate knowing what other doctors are advising. I don’t know if I should use it tonight, or not. Seems like “damned if you do, damned if you don’t”.
Posted on July 26, 2021 at 4:26 pm
Richard says:
I’ve been using Phillips bi-pap 3 years. I have developed serious respiratory problems in the past 6 months which require oxygen 24/7 at 10 liters per minute. I’m a non smoker and have no history of exposures. My physicians cannot determine cause, and are calling it incurable pulmonary fibrosis.
Posted on August 3, 2021 at 12:06 pm
Mike says:
After following instructions on the Philips website, after Consulting with my wife’s sleep doctor, and after Consulting with my wife’s durable goods provider, on or around June 19th we stopped using my wife’s Phillips dream station SUV. On June 24th my wife had respiratory failure and was admitted to the hospital where she stayed until July 2nd.
Posted on August 6, 2021 at 9:46 am
Annie says:
I have been using the Remstar SE for years. I have a persistent cough and nasal congestion/drip. I have recently been diagnosed with breast cancer. I still have not been notified of the recall.
Posted on October 8, 2021 at 11:19 am
Lee says:
I was recently diagnosed with papillary thyroid carcinoma, “Thyroid Cancer”. I was also contacted by Philips via a letter for the recall of my machine and after contacting them via phone it was confirmed that my CPAP machine was on the recall list and given a confirmation number. However I have not been given instructions on what to do.
Posted on October 8, 2021 at 12:57 pm
Deborah says:
Short of breath headach a lot sick stomach eyes irritated shivers weakNess sinus problems
Posted on November 11, 2021 at 4:18 pm
Colleen says:
I’m pissed that we got a bill to pay the c-pap a machine we can use. We only had it about a month. And our own pulimary doctors office can not get any info from Philips. I have sat here day after day for 5 months waiting to hear something. All I have done is register both machines.
Posted on November 22, 2021 at 2:41 pm
Mike says:
used machine for several years And been having high pitched noise in my ears dry throat coughing up mucus every morning put on oxygen and put on dialysis within last couple years
Posted on December 6, 2021 at 12:26 am
Gary says:
Cpap mash re call
Posted on December 27, 2021 at 10:07 pm
dave says:
i have and been using a dream station machine for years. i do have some breathing issues just don’t know what to do from here
Posted on February 4, 2022 at 3:17 pm
Kate says:
If i return a recalled cpap and develop lung problems in the future due to use of a faulty one, how do i prove that if need be later on?
Posted on December 11, 2023 at 5:13 pm
John says:
I have a cpap that is affected but actually didn’t find out about the recall until just yesterday, 1/31/2024.
Posted on February 1, 2024 at 10:55 am